Metformin targets c-MYC oncogene to prevent prostate cancer
- PMID: 24130167
- PMCID: PMC3845895
- DOI: 10.1093/carcin/bgt307
Metformin targets c-MYC oncogene to prevent prostate cancer
Abstract
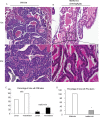
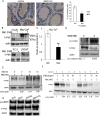
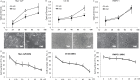
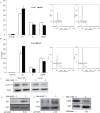
Prostate cancer (PCa) is the second leading cause of cancer-related death in American men and many PCa patients develop skeletal metastasis. Current treatment modalities for metastatic PCa are mostly palliative with poor prognosis. Epidemiological studies indicated that patients receiving the diabetic drug metformin have lower PCa risk and better prognosis, suggesting that metformin may have antineoplastic effects. The mechanism by which metformin acts as chemopreventive agent to impede PCa initiation and progression is unknown. The amplification of c-MYC oncogene plays a key role in early prostate epithelia cell transformation and PCa growth. The purpose of this study is to investigate the effect of metformin on c-myc expression and PCa progression. Our results demonstrated that (i) in Hi-Myc mice that display murine prostate neoplasia and highly resemble the progression of human prostate tumors, metformin attenuated the development of prostate intraepithelial neoplasia (PIN, the precancerous lesion of prostate) and PCa lesions. (ii) Metformin reduced c-myc protein levels in vivo and in vitro. In Myc-CaP mouse PCa cells, metformin decreased c-myc protein levels by at least 50%. (iii) Metformin selectively inhibited the growth of PCa cells by stimulating cell cycle arrest and apoptosis without affecting the growth of normal prostatic epithelial cells (RWPE-1). (iv) Reduced PIN formation by metformin was associated with reduced levels of androgen receptor and proliferation marker Ki-67 in Hi-Myc mouse prostate glands. Our novel findings suggest that by downregulating c-myc, metformin can act as a chemopreventive agent to restrict prostatic neoplasia initiation and transformation.
Summary: Metformin, an old antidiabetes drug, may inhibit prostate intraepithelial neoplasia transforming to cancer lesion via reducing c-MYC, an 'old' overexpressed oncogene. This study explores chemopreventive efficacy of metformin in prostate cancer and its link to cMYC in vitro and in vivo.
Figures

References
-
- Jemal A., et al. (2010). Cancer statistics, 2010. CA Cancer J. Clin., 60, 277–300 - PubMed
-
- SEER (2012). SEER Stat Fact Sheets: Prostate http://seer.cancer.gov/statfacts/html/prost.html (31 January 2013, date last accessed).
-
- Moser L., et al. (2008). Hormone-refractory and metastatic prostate cancer—palliative radiotherapy. Front. Radiat. Ther. Oncol., 41, 117–125 - PubMed
-
- Bostwick D.G. (1988). Premalignant lesions of the prostate. Semin. Diagn. Pathol., 5, 240–253 - PubMed
-
- Colanzi P., et al. (1998). Prostatic intraepithelial neoplasia and prostate cancer: analytical evaluation. Adv. Clin. Path., 2, 271–284 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

