Intravital imaging of hair-cell development and regeneration in the zebrafish
- PMID: 24130521
- PMCID: PMC3795300
- DOI: 10.3389/fnana.2013.00033
Intravital imaging of hair-cell development and regeneration in the zebrafish
Abstract
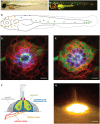
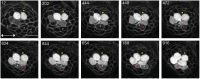
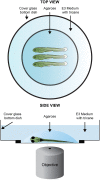
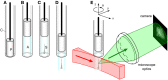
Direct videomicroscopic visualization of organ formation and regeneration in toto is a powerful strategy to study cellular processes that often cannot be replicated in vitro. Intravital imaging aims at quantifying changes in tissue architecture or subcellular organization over time during organ development, regeneration or degeneration. A general feature of this approach is its reliance on the optical isolation of defined cell types in the whole animals by transgenic expression of fluorescent markers. Here we describe a simple and robust method to analyze sensory hair-cell development and regeneration in the zebrafish lateral line by high-resolution intravital imaging using laser-scanning confocal microscopy (LSCM) and selective plane illumination microscopy (SPIM). The main advantage of studying hair-cell regeneration in the lateral line is that it occurs throughout the life of the animal, which allows its study in the most natural context. We detail protocols to achieve continuous videomicroscopy for up to 68 hours, enabling direct observation of cellular behavior, which can provide a sensitive assay for the quantitative classification of cellular phenotypes and cell-lineage reconstruction. Modifications to this protocol should facilitate pharmacogenetic assays to identify or validate otoprotective or reparative drugs for future clinical strategies aimed at preserving aural function in humans.
Keywords: auditory; development; hair cells; intravital fluorescence microscopy; lateral line system; regeneration; zebrafish.
Figures

Similar articles
-
Dynamic neuroanatomy at subcellular resolution in the zebrafish.Methods Mol Biol. 2014;1082:187-95. doi: 10.1007/978-1-62703-655-9_13. Methods Mol Biol. 2014. PMID: 24048935
-
Imaging collective cell migration and hair cell regeneration in the sensory lateral line.Methods Cell Biol. 2016;134:211-56. doi: 10.1016/bs.mcb.2016.01.004. Epub 2016 Feb 28. Methods Cell Biol. 2016. PMID: 27312495
-
Confocal Microscope-Based Laser Ablation and Regeneration Assay in Zebrafish Interneuromast Cells.J Vis Exp. 2020 May 20;(159):10.3791/60966. doi: 10.3791/60966. J Vis Exp. 2020. PMID: 32510477 Free PMC article.
-
Digitizing life at the level of the cell: high-performance laser-scanning microscopy and image analysis for in toto imaging of development.Mech Dev. 2003 Nov;120(11):1407-20. doi: 10.1016/j.mod.2003.07.005. Mech Dev. 2003. PMID: 14623446 Review.
-
Sensory hair cell death and regeneration in fishes.Front Cell Neurosci. 2015 Apr 21;9:131. doi: 10.3389/fncel.2015.00131. eCollection 2015. Front Cell Neurosci. 2015. PMID: 25954154 Free PMC article. Review.
Cited by
-
Inexhaustible hair-cell regeneration in young and aged zebrafish.Biol Open. 2015 May 22;4(7):903-9. doi: 10.1242/bio.012112. Biol Open. 2015. PMID: 26002931 Free PMC article.
-
Evaluation of the Hair Cell Regeneration in Zebrafish Larvae by Measuring and Quantifying the Startle Responses.Neural Plast. 2017;2017:8283075. doi: 10.1155/2017/8283075. Epub 2017 Jan 29. Neural Plast. 2017. PMID: 28250994 Free PMC article.
-
Quantitative videomicroscopy reveals latent control of cell-pair rotations in vivo.Development. 2023 May 1;150(9):dev200975. doi: 10.1242/dev.200975. Epub 2023 May 3. Development. 2023. PMID: 36946430 Free PMC article.
-
Multiphoton intravital microscopy of rodents.Nat Rev Methods Primers. 2022;2:89. doi: 10.1038/s43586-022-00168-w. Epub 2022 Nov 10. Nat Rev Methods Primers. 2022. PMID: 37621948 Free PMC article.
-
Behavioral methods for the functional assessment of hair cells in zebrafish.Front Med. 2017 Jun;11(2):178-190. doi: 10.1007/s11684-017-0507-x. Epub 2017 Mar 27. Front Med. 2017. PMID: 28349300 Review.
References
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

