Rapid responses to reverse T₃ hormone in immature rat Sertoli cells: calcium uptake and exocytosis mediated by integrin
- PMID: 24130850
- PMCID: PMC3795021
- DOI: 10.1371/journal.pone.0077176
Rapid responses to reverse T₃ hormone in immature rat Sertoli cells: calcium uptake and exocytosis mediated by integrin
Abstract
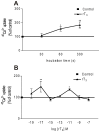
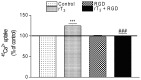
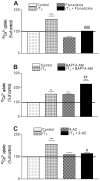
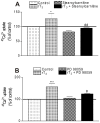
There is increasing experimental evidence of the nongenomic action of thyroid hormones mediated by receptors located in the plasma membrane or inside cells. The aim of this work was to characterize the reverse T₃ (rT₃) action on calcium uptake and its involvement in immature rat Sertoli cell secretion. The results presented herein show that very low concentrations of rT₃ are able to increase calcium uptake after 1 min of exposure. The implication of T-type voltage-dependent calcium channels and chloride channels in the effect of rT₃ was evidenced using flunarizine and 9-anthracene, respectively. Also, the rT₃-induced calcium uptake was blocked in the presence of the RGD peptide (an inhibitor of integrin-ligand interactions). Therefore, our findings suggest that calcium uptake stimulated by rT₃ may be mediated by integrin αvβ₃. In addition, it was demonstrated that calcium uptake stimulated by rT₃ is PKC and ERK-dependent. Furthermore, the outcomes indicate that rT₃ also stimulates cellular secretion since the cells manifested a loss of fluorescence after 4 min incubation, indicating an exocytic quinacrine release that seems to be mediated by the integrin receptor. These findings indicate that rT₃ modulates the calcium entry and cellular secretion, which might play a role in the regulation of a plethora of intracellular processes involved in male reproductive physiology.
Conflict of interest statement
Figures
Similar articles
-
Integrin participates in the effect of thyroxine on plasma membrane in immature rat testis.Biochim Biophys Acta. 2013 Mar;1830(3):2629-37. doi: 10.1016/j.bbagen.2012.10.022. Biochim Biophys Acta. 2013. PMID: 23137442
-
1alpha,25(OH)2-Vitamin D3 stimulation of secretion via chloride channel activation in Sertoli cells.J Steroid Biochem Mol Biol. 2010 Apr;119(3-5):127-34. doi: 10.1016/j.jsbmb.2010.01.011. Epub 2010 Feb 13. J Steroid Biochem Mol Biol. 2010. PMID: 20156558
-
1α,25-Dihydroxyvitamin D(3) signaling pathways on calcium uptake in 30-day-old rat Sertoli cells.Biochemistry. 2011 Nov 29;50(47):10284-92. doi: 10.1021/bi201113n. Epub 2011 Nov 4. Biochemistry. 2011. PMID: 22035182
-
Rapid signaling responses in Sertoli cell membranes induced by follicle stimulating hormone and testosterone: calcium inflow and electrophysiological changes.Life Sci. 2011 Oct 10;89(15-16):577-83. doi: 10.1016/j.lfs.2011.05.017. Epub 2011 Jun 16. Life Sci. 2011. PMID: 21703282 Review.
-
Mechanisms of nongenomic actions of thyroid hormone.Front Neuroendocrinol. 2008 May;29(2):211-8. doi: 10.1016/j.yfrne.2007.09.003. Epub 2007 Oct 5. Front Neuroendocrinol. 2008. PMID: 17983645 Review.
Cited by
-
Molecular Mechanisms and Signaling Pathways Involved in Sertoli Cell Proliferation.Front Endocrinol (Lausanne). 2019 Apr 16;10:224. doi: 10.3389/fendo.2019.00224. eCollection 2019. Front Endocrinol (Lausanne). 2019. PMID: 31040821 Free PMC article. Review.
-
Thyroid hormone and leptin in the testis.Front Endocrinol (Lausanne). 2014 Nov 25;5:198. doi: 10.3389/fendo.2014.00198. eCollection 2014. Front Endocrinol (Lausanne). 2014. PMID: 25505448 Free PMC article. Review.
-
Clinical Electrophysiology and Mathematical Modeling for Precision Diagnosis of Infertility.Biomedicines. 2025 Jan 21;13(2):250. doi: 10.3390/biomedicines13020250. Biomedicines. 2025. PMID: 40002664 Free PMC article. Review.
-
Pyriproxyfen Disrupts the Ongoing Spermatogenesis Wave in Danio rerio Potentially Mediated by Voltage-Dependent Calcium Channels and Protein Kinase C.J Appl Toxicol. 2025 Sep;45(9):1705-1719. doi: 10.1002/jat.4801. Epub 2025 May 1. J Appl Toxicol. 2025. PMID: 40308215 Free PMC article.
-
Prospects for improving future mental health of children through prenatal maternal micronutrient supplementation in China.Pediatr Investig. 2020 Jun 24;4(2):118-126. doi: 10.1002/ped4.12199. eCollection 2020 Jun. Pediatr Investig. 2020. PMID: 32851355 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

