Physical biology in cancer. 4. Physical cues guide tumor cell adhesion and migration
- PMID: 24133064
- PMCID: PMC3919991
- DOI: 10.1152/ajpcell.00289.2013
Physical biology in cancer. 4. Physical cues guide tumor cell adhesion and migration
Abstract
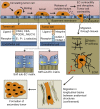
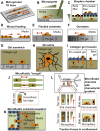
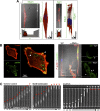
As tumor cells metastasize from the primary tumor location to a distant secondary site, they encounter an array of biologically and physically heterogeneous microenvironments. While it is well established that biochemical signals guide all stages of the metastatic cascade, mounting evidence indicates that physical cues also direct tumor cell behavior, including adhesion and migration phenotypes. Physical cues acting on tumor cells in vivo include extracellular matrix mechanical properties, dimensionality, and topography, as well as interstitial flow, hydrodynamic shear stresses, and local forces due to neighboring cells. State-of-the-art technologies have recently enabled us and other researchers to engineer cell microenvironments that mimic specific physical properties of the cellular milieu. Through integration of these engineering strategies, along with physics, molecular biology, and imaging techniques, we have acquired new insights into tumor cell adhesion and migration mechanisms. In this review, we focus on the extravasation and invasion stages of the metastatic cascade. We first discuss the physical role of the endothelium during tumor cell extravasation and invasion and how contractility of endothelial and tumor cells contributes to the ability of tumor cells to exit the vasculature. Next, we examine how matrix dimensionality and stiffness coregulate tumor cell adhesion and migration beyond the vasculature. Finally, we summarize how tumor cells translate and respond to physical cues through mechanotransduction. Because of the critical role of tumor cell mechanotransduction at various stages of the metastatic cascade, targeting signaling pathways involved in tumor cell mechanosensing of physical stimuli may prove to be an effective therapeutic strategy for cancer patients.
Keywords: cell adhesion; extravasation; matrix stiffness; three-dimensional migration; tumor metastasis.
Figures
References
-
- Al-Mehdi AB, Tozawa K, Fisher AB, Shientag L, Lee A, Muschel RJ. Intravascular origin of metastasis from the proliferation of endothelium-attached tumor cells: a new model for metastasis. Nat Med 6: 100–102, 2000 - PubMed
-
- Balgude AP, Yu X, Szymanski A, Bellamkonda RV. Agarose gel stiffness determines rate of DRG neurite extension in 3D cultures. Biomaterials 22: 1077–1084, 2001 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

