Systematic screen with kinases inhibitors reveals kinases play distinct roles in growth of osteoprogenitor cells
- PMID: 24133586
- PMCID: PMC3796230
Systematic screen with kinases inhibitors reveals kinases play distinct roles in growth of osteoprogenitor cells
Abstract
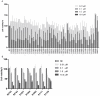
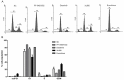
Cancer treatment-related bone loss has become growing problematic, especially in breast and prostate cancer treated with hormone/endocrine therapy, chemotherapy and radiotherapy. However, bone loss caused by targeted therapy in cancer patients is largely unknown yet. In present study, a kinase inhibitors screen was applied for MC3T3-E1, a murine osteoprogenitor cell line, and seven kinase inhibitors (GSK1838705A, PF-04691502, Dasatinib, Masitinib, GDC-0941, XL880 and Everolimus) were found to suppress the cell viability with dose- and time-dependent manner. The most interesting is that many kinase inhibitors (such as lapatinib, erlotinib and sunitinib) can promote MC3T3-E1 cell proliferation at 0.01 μM. 4 out of 7 inhibitors were selected to perform the functional study and found that they lead to cell cycle dysregulation, treatments of PF-04691502 (AKT inhibitor), Dasatinib (Src inhibitor) and Everolimus (mTOR inhibitor) lead to G1 arrest of MC3T3-E1 cells via downregulation of cyclin D1 and p-AKT, whereas XL880 (MET and VEGFR inhibitor) treatment results in increase of sub-G1 and G2/M phase by upregulation of p53 protein. Our work provides important indications for the comprehensive care of cancer patients treated with some targeted drugs.
Keywords: Cancer treatment-related bone loss; kinases inhibitors screening; osteoprogenitor cells.
Figures


Similar articles
-
Antitumor and antiangiogenic effect of the dual EGFR and HER-2 tyrosine kinase inhibitor lapatinib in a lung cancer model.BMC Cancer. 2010 May 11;10:188. doi: 10.1186/1471-2407-10-188. BMC Cancer. 2010. PMID: 20459769 Free PMC article.
-
The investigational Aurora kinase A inhibitor alisertib (MLN8237) induces cell cycle G2/M arrest, apoptosis, and autophagy via p38 MAPK and Akt/mTOR signaling pathways in human breast cancer cells.Drug Des Devel Ther. 2015 Mar 16;9:1627-52. doi: 10.2147/DDDT.S75378. eCollection 2015. Drug Des Devel Ther. 2015. PMID: 25834401 Free PMC article.
-
Treatment of melanoma with selected inhibitors of signaling kinases effectively reduces proliferation and induces expression of cell cycle inhibitors.Med Oncol. 2017 Dec 6;35(1):7. doi: 10.1007/s12032-017-1069-0. Med Oncol. 2017. PMID: 29214525 Free PMC article.
-
The pan-inhibitor of Aurora kinases danusertib induces apoptosis and autophagy and suppresses epithelial-to-mesenchymal transition in human breast cancer cells.Drug Des Devel Ther. 2015 Feb 17;9:1027-62. doi: 10.2147/DDDT.S74412. eCollection 2015. Drug Des Devel Ther. 2015. PMID: 25733818 Free PMC article.
-
Clinical Management of Potential Toxicities and Drug Interactions Related to Cyclin-Dependent Kinase 4/6 Inhibitors in Breast Cancer: Practical Considerations and Recommendations.Oncologist. 2017 Sep;22(9):1039-1048. doi: 10.1634/theoncologist.2017-0142. Epub 2017 Jul 13. Oncologist. 2017. PMID: 28706010 Free PMC article. Review.
Cited by
-
Receptor tyrosine kinases: Characterisation, mechanism of action and therapeutic interests for bone cancers.J Bone Oncol. 2015 Jan 23;4(1):1-12. doi: 10.1016/j.jbo.2015.01.001. eCollection 2015 Mar. J Bone Oncol. 2015. PMID: 26579483 Free PMC article. Review.
-
GSK1838705A, an IGF-1R inhibitor, inhibits glioma cell proliferation and suppresses tumor growth in vivo.Mol Med Rep. 2015 Oct;12(4):5641-6. doi: 10.3892/mmr.2015.4129. Epub 2015 Jul 28. Mol Med Rep. 2015. PMID: 26238593 Free PMC article.
References
-
- Coleman RE, Rathbone E, Brown JE. Management of cancer treatment-induced bone loss. Nat Rev Rheumatol. 2013;9:365–374. - PubMed
-
- Hershman D, Narayanan R. Prevention and management of osteoporosis in women with breast cancer and men with prostate cancer. Curr Oncol Rep. 2004;6:277–284. - PubMed
-
- Hofbauer LC, Schoppet M. Clinical implications of the osteoprotegerin/RANKL/RANK system for bone and vascular diseases. JAMA. 2004;292:490–495. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
