Mass spectrometry imaging for dissecting steroid intracrinology within target tissues
- PMID: 24134553
- PMCID: PMC4392804
- DOI: 10.1021/ac402777k
Mass spectrometry imaging for dissecting steroid intracrinology within target tissues
Abstract
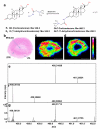
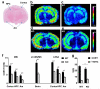
Steroid concentrations within tissues are modulated by intracellular enzymes. Such "steroid intracrinology" influences hormone-dependent cancers and obesity and provides targets for pharmacological inhibition. However, no high resolution methods exist to quantify steroids within target tissues. We developed mass spectrometry imaging (MSI), combining matrix assisted laser desorption ionization with on-tissue derivatization with Girard T and Fourier transform ion cyclotron resonance mass spectrometry, to quantify substrate and product (11-dehydrocorticosterone and corticosterone) of the glucocorticoid-amplifying enzyme 11β-HSD1. Regional steroid distribution was imaged at 150-200 μm resolution in rat adrenal gland and mouse brain sections and confirmed with collision induced dissociation/liquid extraction surface analysis. In brains of mice with 11β-HSD1 deficiency or inhibition, MSI quantified changes in subregional corticosterone/11-dehydrocorticosterone ratio, distribution of inhibitor, and accumulation of the alternative 11β-HSD1 substrate, 7-ketocholesterol. MSI data correlated well with LC-MS/MS in whole brain homogenates. MSI with derivatization is a powerful new tool to investigate steroid biology within tissues.
Figures


References
-
- Simpson ER, Mahendroo MS, Means GD, Kilgore MW, Hinshelwood MM, Graham-Lorence S, Amareh B, Ito Y, Fisher CR, Michael MD, Mendelson CR, Bulun SE. Endocr. Rev. 1994;15:342–355. - PubMed
-
- Russell DW, Wilson JD. Annual Reviews of Biochemistry. 1994;63:25–61. - PubMed
-
- Seckl JR, Walker BR. TEM. 2004;15:418–424. - PubMed
-
- Malmberg H, Nygren P, Sjövall L. a. J. L. Spectroscopy. 2013;18:503–511.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

