Met activation is required for early cytoprotection after ischemic kidney injury
- PMID: 24136921
- PMCID: PMC3904569
- DOI: 10.1681/ASN.2013050473
Met activation is required for early cytoprotection after ischemic kidney injury
Abstract
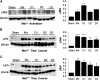
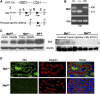
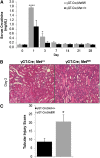
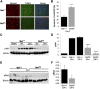
Renal proximal tubule epithelial cells express high levels of the hepatocyte growth factor receptor Met, and both the receptor and ligand are upregulated after ischemic injury. Activation of the Met receptor after hepatocyte growth factor stimulation in vitro promotes activities involved in kidney repair, including cell survival, migration, and proliferation. However, characterizing the in vivo role of these signaling events in proximal tubule responses to kidney injury has been difficult because global Met knockout results in embryonic lethality due to placental and liver abnormalities. Here, we used γGT-Cre to knockout Met receptor expression selectively in the proximal tubules of mice (γGT-Cre;Met(fl/fl)). The kidneys of these mice developed normally, but exhibited increased initial tubular injury, tubular cell apoptosis, and serum creatinine after ischemia/reperfusion compared with γGT-Cre;Met(+/+) kidneys. These changes in γGT-Cre;Met(fl/fl) mice correlated with a selective reduction in PI3K/Akt activation in response to injury and subsequent decreases in inhibitory phosphorylation of the proapoptotic factor Bad and activating phosphorylation of the ribosomal regulatory protein p70-S6 kinase. Moreover, tubular cell proliferation after ischemia/reperfusion was delayed in γGT-Cre;Met(fl/fl) mice. In conclusion, this study identifies Met-dependent phosphoinositide 3-kinase activation in proximal tubules as a critical determinant of initial tubular cell survival and reparative proliferation after ischemic injury.
Figures
Similar articles
-
Deletion of the epidermal growth factor receptor in renal proximal tubule epithelial cells delays recovery from acute kidney injury.Kidney Int. 2012 Jul;82(1):45-52. doi: 10.1038/ki.2012.43. Epub 2012 Mar 14. Kidney Int. 2012. PMID: 22418982 Free PMC article.
-
Focal adhesion kinase signaling mediates acute renal injury induced by ischemia/reperfusion.Am J Pathol. 2011 Dec;179(6):2766-78. doi: 10.1016/j.ajpath.2011.08.025. Epub 2011 Oct 5. Am J Pathol. 2011. PMID: 21982831 Free PMC article.
-
Divergent roles for kidney proximal tubule and granulocyte PAD4 in ischemic AKI.Am J Physiol Renal Physiol. 2018 May 1;314(5):F809-F819. doi: 10.1152/ajprenal.00569.2017. Epub 2018 Jan 3. Am J Physiol Renal Physiol. 2018. PMID: 29357426 Free PMC article.
-
Previous ischemia and reperfusion injury results in resistance of the kidney against subsequent ischemia and reperfusion insult in mice; a role for the Akt signal pathway.Nephrol Dial Transplant. 2012 Oct;27(10):3762-70. doi: 10.1093/ndt/gfs097. Epub 2012 May 3. Nephrol Dial Transplant. 2012. PMID: 22555250
-
Kidney Proximal Tubular TLR9 Exacerbates Ischemic Acute Kidney Injury.J Immunol. 2018 Aug 1;201(3):1073-1085. doi: 10.4049/jimmunol.1800211. Epub 2018 Jun 13. J Immunol. 2018. PMID: 29898963 Free PMC article.
Cited by
-
Suppressed autophagic response underlies augmentation of renal ischemia/reperfusion injury by type 2 diabetes.Sci Rep. 2017 Jul 13;7(1):5311. doi: 10.1038/s41598-017-05667-5. Sci Rep. 2017. PMID: 28706237 Free PMC article.
-
Protective effect of luteolin on skin ischemia-reperfusion injury through an AKT-dependent mechanism.Int J Mol Med. 2018 Dec;42(6):3073-3082. doi: 10.3892/ijmm.2018.3915. Epub 2018 Oct 2. Int J Mol Med. 2018. PMID: 30280183 Free PMC article.
-
Three-Dimensional Morphology by Multiphoton Microscopy with Clearing in a Model of Cisplatin-Induced CKD.J Am Soc Nephrol. 2016 Apr;27(4):1102-12. doi: 10.1681/ASN.2015010079. Epub 2015 Aug 24. J Am Soc Nephrol. 2016. PMID: 26303068 Free PMC article.
-
Chemokine receptor Cxcr4 contributes to kidney fibrosis via multiple effectors.Am J Physiol Renal Physiol. 2015 Mar 1;308(5):F459-72. doi: 10.1152/ajprenal.00146.2014. Epub 2014 Dec 23. Am J Physiol Renal Physiol. 2015. PMID: 25537742 Free PMC article.
-
The Circular RNA ciRs-126 Predicts Survival in Critically Ill Patients With Acute Kidney Injury.Kidney Int Rep. 2018 Jun 2;3(5):1144-1152. doi: 10.1016/j.ekir.2018.05.012. eCollection 2018 Sep. Kidney Int Rep. 2018. PMID: 30197981 Free PMC article.
References
-
- Brezis M, Rosen S: Hypoxia of the renal medulla—its implications for disease. N Engl J Med 332: 647–655, 1995 - PubMed
-
- Chou SY, Porush JG, Faubert PF: Renal medullary circulation: Hormonal control. Kidney Int 37: 1–13, 1990 - PubMed
-
- Bonventre JV: Mechanisms of ischemic acute renal failure. Kidney Int 43: 1160–1178, 1993 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

