A canonical to non-canonical Wnt signalling switch in haematopoietic stem-cell ageing
- PMID: 24141946
- PMCID: PMC4078992
- DOI: 10.1038/nature12631
A canonical to non-canonical Wnt signalling switch in haematopoietic stem-cell ageing
Abstract
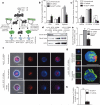
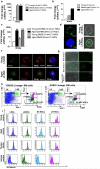
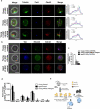
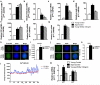
Many organs with a high cell turnover (for example, skin, intestine and blood) are composed of short-lived cells that require continuous replenishment by somatic stem cells. Ageing results in the inability of these tissues to maintain homeostasis and it is believed that somatic stem-cell ageing is one underlying cause of tissue attrition with age or age-related diseases. Ageing of haematopoietic stem cells (HSCs) is associated with impaired haematopoiesis in the elderly. Despite a large amount of data describing the decline of HSC function on ageing, the molecular mechanisms of this process remain largely unknown, which precludes rational approaches to attenuate stem-cell ageing. Here we report an unexpected shift from canonical to non-canonical Wnt signalling in mice due to elevated expression of Wnt5a in aged HSCs, which causes stem-cell ageing. Wnt5a treatment of young HSCs induces ageing-associated stem-cell apolarity, reduction of regenerative capacity and an ageing-like myeloid-lymphoid differentiation skewing via activation of the small Rho GTPase Cdc42. Conversely, Wnt5a haploinsufficiency attenuates HSC ageing, whereas stem-cell-intrinsic reduction of Wnt5a expression results in functionally rejuvenated aged HSCs. Our data demonstrate a critical role for stem-cell-intrinsic non-canonical Wnt5a signalling in HSC ageing.
Figures





Comment in
-
Stem cells: a WNT switch to ageing.Nat Rev Mol Cell Biol. 2013 Dec;14(12):752. doi: 10.1038/nrm3701. Epub 2013 Nov 8. Nat Rev Mol Cell Biol. 2013. PMID: 24201108 No abstract available.
-
Noncanonical Wnt comes of age in hematopoietic stem cells.Cell Stem Cell. 2013 Dec 5;13(6):642-3. doi: 10.1016/j.stem.2013.11.009. Cell Stem Cell. 2013. PMID: 24315437
References
-
- Morrison SJ, Uchida N, Weissman IL. The biology of hematopoieticstem cells. Annu. Rev. Cell Dev. Biol. 1995;11:35–71. - PubMed
-
- Fuchs E, Segre JA. Stem cells: a new lease on life. Cell. 2000;100:143–155. - PubMed
-
- Rossi DJ, Jamieson CH, Weissman IL. Stems cells and the pathways to aging and cancer. Cell. 2008;132:681–696. - PubMed
-
- Geiger H, Van Zant G. The aging of lympho-hematopoietic stem cells. Nature Immunol. 2002;3:329–333. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

