CCR2(+) monocytes infiltrate atrophic lesions in age-related macular disease and mediate photoreceptor degeneration in experimental subretinal inflammation in Cx3cr1 deficient mice
- PMID: 24142887
- PMCID: PMC3840491
- DOI: 10.1002/emmm.201302692
CCR2(+) monocytes infiltrate atrophic lesions in age-related macular disease and mediate photoreceptor degeneration in experimental subretinal inflammation in Cx3cr1 deficient mice
Abstract
Atrophic age-related macular degeneration (AMD) is associated with the subretinal accumulation of mononuclear phagocytes (MPs). Their role in promoting or inhibiting retinal degeneration is unknown. We here show that atrophic AMD is associated with increased intraocular CCL2 levels and subretinal CCR2(+) inflammatory monocyte infiltration in patients. Using age- and light-induced subretinal inflammation and photoreceptor degeneration in Cx3cr1 knockout mice, we show that subretinal Cx3cr1 deficient MPs overexpress CCL2 and that both the genetic deletion of CCL2 or CCR2 and the pharmacological inhibition of CCR2 prevent inflammatory monocyte recruitment, MP accumulation and photoreceptor degeneration in vivo. Our study shows that contrary to CCR2 and CCL2, CX3CR1 is constitutively expressed in the retina where it represses the expression of CCL2 and the recruitment of neurotoxic inflammatory CCR2(+) monocytes. CCL2/CCR2 inhibition might represent a powerful tool for controlling inflammation and neurodegeneration in AMD.
Keywords: age-related macular disease; chemokines; monocyte; neurodegeneration; neuroinflammation.
© 2013 The Authors. Published by John Wiley and Sons, Ltd on behalf of EMBO.
Figures
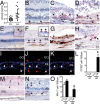
A. CCL2 ELISA of aqueous humours of geographic atrophy (GA) patients and control subjects (n = 18 GA patients n = 22 control patients, student t-test p < 0.0001; Mann–Whitney test p < 0.0001).
B–D. CCL2 immunohistochemistry (red staining) on macular sections of (B) control donor tissues, (C and D) within the GA lesion.
E–H. CCR2 immunohistochemistry (red staining) on macular sections of (E) control donor tissues, inset: major retinal vessel containing erythrocytes and leucocytes, (F) within the GA lesion, inset: adjacent to GA lesion (G) laminar deposit (H) soft drusen.
I–L. (I) CCR2 (green staining), (J) CD18 (red staining), (K) merge double labelling of a GA lesion. (L) Quantification of intraretinal and subretinal CCR2/CD18 positive cells expressed as CCR2+ cells/500 mm of the atrophic lesion (n = 10 GA donor maculae from 7 patients: age, mean (SD): 84 (8.8) and n = 5 control maculae from 5 patients: age, mean (SD): 83 (8.8), *students t-test p = 0.001).
M, N. IBA-1 immunohistochemistry (red staining) on macular sections of (M) control donor tissues, (N) within the GA lesion.
O. (O) Quantification of intraretinal and subretinal IBA-1 positive cells expressed as IBA-1+ cells/500 mm of the atrophic lesion (subjects same as above, *students t-test p = 0.005). B–L and N and O: representative images from 5 healthy donors, 7 donors (10 eyes) with GA and 3 donors with age related maculopathy (4 eyes), controls omitting the primary antibody showed no staining. All values are represented as mean ± SEM. CTL: control; GA: geographic atrophy; ONL: outer nuclear layer; INL: inner nuclear layer; Ch: choroid; iR: inner retina; sR: subretinal; Scale bar B–J = 50 μm.

A. Quantitative RT-PCR of Ccl2 mRNA normalized with β-actin mRNA of 2- and 18-month-old C57BL/6 and Cx3cr1−/− mouse retina (n = 4 per group, *two-way Anova, Bonferroni p < 0.001).
B. Quantitative RT-PCR of Ccl2 mRNA normalized with β-actin mRNA of non-injured (NI) and at day 4 and day 14 of the light-challenge model of 2- to 3-month-old C57BL/6 and Cx3cr1−/− mice (n = 5–6 per group, *two-way Anova, Bonferroni p < 0,05).
C. CCL2 ELISA protein quantification of retinal protein extracts from non-injured (NI) and at day 14 (d14) of the light-challenge model of 2- to 3-month-old C57BL/6 and Cx3cr1−/− mice (expressed as pg/mg total retinal protein; n = 4 per group, *two-way Anova, Bonferroni p < 0.001).
D–G. Immunohistochemistry CCL2 (red; arrows), and IBA-1 (green; arrows colocalization yellow) of the subretinal side (D and E) and vitreal aspect (F and G) of a retinal flatmount from a Cx3cr1−/− mouse at day 14 (d14) of the light-challenge model (representative of 3 independent experiments, immunostaining omitting the primary antibody or performed on Ccl2−/− mice served as negative controls).
H. Quantitative RT-PCR of Ccl2 mRNA normalized with β-actin mRNA of 2- to 3-month-old C57BL/6, Ccl2−/−, Ccr2−/−, Cx3cr1GFP/GFP and Cx3cr1GFP/GFPCcr2RFP/RFP mice at day 4 of the light-challenge model (d4) (n = 5–6 per group, *two-way Anova, Bonferroni p < 0.01).
I. Quantitative RT-PCR of Ccl2 mRNA normalized with S26 of whole-eye-lysats (set as 1) and FACS-sorted GFPhighLy6Cneg and GFPlowLy6Chigh cells pooled from 8 eyes of PBS perfused Cx3cr1GFP/GFP mice after 4 days of light challenge.
J. (J) Quantitative RT-PCR of Ccl2 mRNA normalized with S26 mRNA of Cx3cr1+/+ and Cx3cr1−/− monocyte derived Mφs cultivated for 18 h with or without photoreceptor outer segments (POSs, n = 4 per group, two-way Anova Bonferroni *p < 0.001). All values are represented as mean ± SEM. Scale bar D–G = 50 μm; CTL: control; +POS: +photoreceptor outer segments.
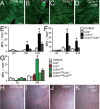
A–D. 12 month-old IBA-1 stained RPE-flatmounts of C57BL/6 (A), Ccl2−/− (B), Cx3cr1−/− (C) and Cx3cr1−/−Ccl2−/− (D).
E. Quantification of subretinal MPs/mm2 at 3, 9 and 12 months (n = 6 per group, *two-way Anova Bonferroni p < 0,001).
F. Quantification of subretinal MPs/mm2 non-injured and at day 4 and 14 of the light-challenge model of 2- to 3-month-old C57BL/6J, Ccl2−/−, Cx3cr1−/− and Cx3cr1−/−Ccl2−/− mice (n = 6–9 per group, *two-way Anova Bonferroni p < 0.001).
G. Quantification of IBA-1 positive subretinal MPs/mm2 in non-injured and at day 14 of the light-challenge model of 2- to 3-month-old C57BL/6J, Ccl2−/−, CCR2−/−, Cx3cr1GFP/GFP, Cx3cr1GFP/GFPCcl2−/− and Cx3cr1GFP/GFPCcr2RFP/RFP mice (n = 6–9 per group, *two-way Anova Bonferroni p < 0.001).
H–K. Representative fundoscopic photographs of 12-month-old C57BL/6 (H), Ccl2−/− (I), Cx3cr1−/− (J) and Cx3cr1−/−Ccl2−/− (K) (n > 8 per group). All values are represented as mean ± SEM. Scale A–D = 100 μm.

A–D. Micrographs, taken 1000 μm from the optic nerve of 12-month-old C57BL/6J (A), Ccl2−/− (B), Cx3cr1−/− (C, arrows: photoreceptor nuclei; star: nucleus of a subretinal macrophage) and Cx3cr1−/−Ccl2−/− (D).
E–G. (E) Photoreceptor nucleus rows at increasing distances (−3000 μm: inferior pole, +3000 μm: superior pole) from the optic nerve (0 μm) in 12-month-old C57BL/6J, Ccl2−/−, Cx3cr1−/− and Cx3cr1−/−Ccl2−/− mice (n = 4–7, the area under the curve in Cx3cr1−/− mice tested significantly different from the other strains *one-way Anova Bonferroni p < 0.05). Hoechst (blue staining), TUNEL (red staining), rhodopsin (green staining) labelling of Cx3cr1−/− mice at d14 of the light-challenge model in sections (F inset showing rhodopsin double labelling) and confocal microscopy of flatmounts with z-stack projections (G).
H–K. TUNEL stained retinal flatmounts of d14 light-challenged C57BL/6 (H), Ccl2−/− (I), Cx3cr1−/− (J) and Cx3cr1−/−Ccl2−/−(K) mice.
L. Quantification of TUNEL positive cells per retina (n = 4–6 per group, *one-way Anova Bonferroni p < 0.01). All values are represented as mean ± SEM. ONL: outer nuclear layer. Scale bars: A–D and G = 50 μm; F and H–K = 100 μm.
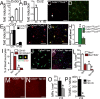
A, B. RT-PCR of relative (A) Cx3cr1 and (B) Ccr2 mRNA expression normalized with S26 mRNA in C57BL/6 blood monocytes (Mo), retina (R) and retinal microglial cells (MC) (n = 3 independent cell preparations, all groups significantly different from each other for Cx3cr1 by one-way Anova Bonferoni p < 0.05; Mo significantly different from MC and R for Ccr2 by two-way Anova Bonferoni p < 0.001).
C, D. Confocal microscopy of (C) RFP fluorescence and (D) merged RFP and GFP fluorescence of the outer plexiform layer of non-injured Cx3cr1+/GFPCcr2+/RFPmice.
E. Quantitative RT-PCR of Ccr2 mRNA normalized with S26 mRNA of Cx3cr1+/+ and Cx3cr1−/− freshly prepared monocytes and monocyte derived Mφs cultivated for 18 h in direct contact with photoreceptor outer segments (POS) (n = 4 per group, two-way Anova Bonferroni p < 0.001 significant difference in monocytes compared to Mφs; no difference between genotypes).
F, G. (F) RFP fluorescence and (G) merged RFP and GFP fluorescence of Hoechst stained chroidal/RPE flatmount after 4 days of light-challenge of Cx3cr1GFP/GFPCcr2+/RFP mice.
H. Quantification of RFP positive subretinal cells after 4 days of light-challenge in Cx3cr1+/GFPCcr2+/RFP, Cx3cr1GFP/GFPCcr2+/RFP and Cx3cr1GFP/GFPCcr2RFP/RFP mice (n = 4 per group, *one-way Anova Bonferroni p < 0.01).
I. Quantification of the percentage of CD115+ (inset green staining) and CD115+EdU+ cells (inset red/green staining) of all Hoechst positive leukocytes of blood smears from day 4 light-challenged Cx3cr1−/− mice that all received three daily intraperitoneal EdU injections and one daily intravenous injection of either control or clodronate liposome (n = 4 per group, two-way Anova Bonferroni p < 0.001 significant difference in the number of CD115+ and CD115+EdU+ cells in clodronate liposome treated animals compared to controls).
J, K. IBA-1 (green) EdU (red) double labelled chroidal/RPE flatmount of intraperitoneally EdU-injected light-challenged Cx3cr1−/− mice receiving control liposome injections (J) or clodronate liposome injections (K).
L. Quantification of subretinal IBA-1+ and IBA-1+EdU+ cells of control liposome and clodronate liposomes treated 4 days light-challenged Cx3cr1−/− mice (n = 4 mice per group, two-way Anova Bonferroni p < 0.001 significant difference in the numbers of subretinal *IBA-1+ MPs and †IBA-1+EdU+ MPs between clodronate liposome and control liposome treated mice).
M, N. TUNEL stained retinal-flatmounts of control liposome (M) and clodronate liposome (N) treated light-challenged Cx3cr1−/− at d14.
O, P. O) Quantification of IBA-1 positive subretinal MPs/mm2 (n = 4–7 per group, *Mann–Whitney p = 0.03) and (P) TUNEL positive photoreceptor nuclei in control liposome and clodronate liposome treated light-challenged Cx3cr1−/− at d14 (n = 4–7 * Mann–Whitney p < 0.0001). All values are represented as mean ± SEM. R: Retina; MC: microglial cells; Mo: monocytes; POS: photoreceptor outer segments; lipo: empty control liposomes; lipo-clo: clodronate liposomes. Scale bars = 50 μm.

A–C. Confocal microscopy of flatmounts with z-stack projections (Hoechst in blue) of TUNEL (red) stained retinal-flatmounts of cultured for 18 h without MPs (A), in contact with C57BL/6 monocytes (B) and Cx3cr1−/− monocytes (C).
D. Quantification of TUNEL+nuclei/mm2 in the ONL of the different groups (n = 8 per group, one-way Anova Bonferroni *significant difference between explants without MPs and C57BL/6 monocytes p < 0.01; † significant difference between explants with C57BL/6 monocytes and Cx3cr1−/− monocytes p < 0.01; $ significant difference between Cx3cr1−/− monocytes and Cx3cr1−/− MC p < 0.001). All values are represented as mean ± SEM. Mo: monocytes; MC: microglial cells. Scale bar = 50 μm.
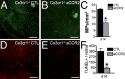
A, B. IBA-1 stained RPE-flatmounts of PBS (A) and CCR2 inhibitor RS 102895 (B) treated light-challenged Cx3cr1−/− mice at d14.
C–E. C) Quantification of IBA-1 positive subretinal MPs/mm2 (n = 6–10 per group, *Mann–Whitney p = 0.01). TUNEL stained retinal-flatmounts of PBS (D) and CCR2 inhibitor (E) treated light-challenged Cx3cr1−/− mice at d14.
F. Quantification of TUNEL positive photoreceptor nuclei per retina (n = 4 per group, *Mann–Whitney p = 0.028). All values are represented as mean ± SEM. Scale bar = 100 μm.
Similar articles
-
CCL2/CCR2 and CX3CL1/CX3CR1 chemokine axes and their possible involvement in age-related macular degeneration.J Neuroinflammation. 2010 Dec 2;7:87. doi: 10.1186/1742-2094-7-87. J Neuroinflammation. 2010. PMID: 21126357 Free PMC article. Review.
-
APOE Isoforms Control Pathogenic Subretinal Inflammation in Age-Related Macular Degeneration.J Neurosci. 2015 Oct 7;35(40):13568-76. doi: 10.1523/JNEUROSCI.2468-15.2015. J Neurosci. 2015. PMID: 26446211 Free PMC article.
-
Ccl2, Cx3cr1 and Ccl2/Cx3cr1 chemokine deficiencies are not sufficient to cause age-related retinal degeneration.Exp Eye Res. 2013 Feb;107:80-7. doi: 10.1016/j.exer.2012.11.015. Epub 2012 Dec 8. Exp Eye Res. 2013. PMID: 23232206 Free PMC article.
-
Upregulation of P2RX7 in Cx3cr1-Deficient Mononuclear Phagocytes Leads to Increased Interleukin-1β Secretion and Photoreceptor Neurodegeneration.J Neurosci. 2015 May 6;35(18):6987-96. doi: 10.1523/JNEUROSCI.3955-14.2015. J Neurosci. 2015. PMID: 25948251 Free PMC article.
-
Local vs. systemic mononuclear phagocytes in age-related macular degeneration and their regulation by CCL2-CCR2 and CX3CL1-CX3CR1 chemokine signalling.Adv Exp Med Biol. 2012;723:17-22. doi: 10.1007/978-1-4614-0631-0_3. Adv Exp Med Biol. 2012. PMID: 22183310 Review. No abstract available.
Cited by
-
Potential gene identification and pathway crosstalk analysis of age-related macular degeneration.Front Genet. 2022 Sep 6;13:992328. doi: 10.3389/fgene.2022.992328. eCollection 2022. Front Genet. 2022. PMID: 36147504 Free PMC article.
-
Microglia Activation and Inflammation During the Death of Mammalian Photoreceptors.Annu Rev Vis Sci. 2020 Sep 15;6:149-169. doi: 10.1146/annurev-vision-121219-081730. Annu Rev Vis Sci. 2020. PMID: 32936734 Free PMC article. Review.
-
Characterization and identification of measurable endpoints in a mouse model featuring age-related retinal pathologies: a platform to test therapies.Lab Invest. 2022 Oct;102(10):1132-1142. doi: 10.1038/s41374-022-00795-7. Epub 2022 May 19. Lab Invest. 2022. PMID: 35589984 Free PMC article.
-
VEGF-production by CCR2-dependent macrophages contributes to laser-induced choroidal neovascularization.PLoS One. 2014 Apr 8;9(4):e94313. doi: 10.1371/journal.pone.0094313. eCollection 2014. PLoS One. 2014. PMID: 24714223 Free PMC article.
-
Hyperreflective Foci, Optical Coherence Tomography Progression Indicators in Age-Related Macular Degeneration, Include Transdifferentiated Retinal Pigment Epithelium.Invest Ophthalmol Vis Sci. 2021 Aug 2;62(10):34. doi: 10.1167/iovs.62.10.34. Invest Ophthalmol Vis Sci. 2021. PMID: 34448806 Free PMC article.
References
-
- Ambati J, Anand A, Fernandez S, Sakurai E, Lynn BC, Kuziel WA, Rollins BJ, Ambati BK. An animal model of age-related macular degeneration in senescent Ccl-2- or Ccr-2-deficient mice. Nat Med. 2003;9(11):1390–1397. - PubMed
-
- Anastasopoulos E, Kakoulidou A, Coleman AL, Sinsheimer JS, Wilson MR, Yu F, Salonikiou A, Koskosas A, Pappas T, Founti P, et al. Association of sequence variation in the CX3CR1 gene with geographic atrophy age-related macular degeneration in a Greek population. Curr Eye Res. 2012;37:1148–1155. - PubMed
-
- Bazan JF, Bacon KB, Hardiman G, Wang W, Soo K, Rossi D, Greaves DR, Zlotnik A, Schall TJ. A new class of membrane-bound chemokine with a CX3C motif. Nature. 1997;385:640–644. - PubMed
-
- Cardona AE, Pioro EP, Sasse ME, Kostenko V, Cardona SM, Dijkstra IM, Huang D, Kidd G, Dombrowski S, Dutta R, et al. Control of microglial neurotoxicity by the fractalkine receptor. Nat Neurosci. 2006;9:917–924. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

