Heat shock protein B1-deficient mice display impaired wound healing
- PMID: 24143227
- PMCID: PMC3797036
- DOI: 10.1371/journal.pone.0077383
Heat shock protein B1-deficient mice display impaired wound healing
Abstract
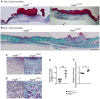
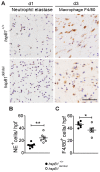
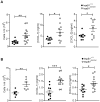
There is large literature describing in vitro experiments on heat shock protein (hsp)B1 but understanding of its function in vivo is limited to studies in mice overexpressing human hspB1 protein. Experiments in cells have shown that hspB1 has chaperone activity, a cytoprotective role, regulates inflammatory gene expression, and drives cell proliferation. To investigate the function of the protein in vivo we generated hspB1-deficient mice. HspB1-deficient fibroblasts display increased expression of the pro-inflammatory cytokine, interleukin-6, compared to wild-type cells, but reduced proliferation. HspB1-deficient fibroblasts exhibit reduced entry into S phase and increased expression of cyclin-dependent kinase inhibitors p27(kip1) and p21(waf1). The expression of hspB1 protein and mRNA is also controlled by the cell cycle. To investigate the physiological function of hspB1 in regulating inflammation and cell proliferation we used an excisional cutaneous wound healing model. There was a significant impairment in the rate of healing of wounds in hspB1-deficient mice, characterised by reduced re-epithelialisation and collagen deposition but also increased inflammation. HspB1 deficiency augments neutrophil infiltration in wounds, driven by increased chemokine (C-X-C motif) ligand 1 expression. This appears to be a general mechanism as similar results were obtained in the air-pouch and peritonitis models of acute inflammation.
Conflict of interest statement
Figures


Similar articles
-
Cardiomyocyte-specific deficiency of HSPB1 worsens cardiac dysfunction by activating NFκB-mediated leucocyte recruitment after myocardial infarction.Cardiovasc Res. 2019 Jan 1;115(1):154-167. doi: 10.1093/cvr/cvy163. Cardiovasc Res. 2019. PMID: 29982352 Free PMC article.
-
Regulation of stress-induced intracellular sorting and chaperone function of Hsp27 (HspB1) in mammalian cells.Biochem J. 2007 Nov 1;407(3):407-17. doi: 10.1042/BJ20070195. Biochem J. 2007. PMID: 17650072 Free PMC article.
-
Heat shock protein 27: induction by gastroduodenal reflux in vivo and augmentation of human esophageal mucosal cell growth in vitro.J Thorac Cardiovasc Surg. 2010 Apr;139(4):1019-25. doi: 10.1016/j.jtcvs.2009.09.066. J Thorac Cardiovasc Surg. 2010. PMID: 20304146
-
HSPB1, actin filament dynamics, and aging cells.Ann N Y Acad Sci. 2010 Jun;1197:76-84. doi: 10.1111/j.1749-6632.2010.05191.x. Ann N Y Acad Sci. 2010. PMID: 20536836 Review.
-
HspB1 (Hsp 27) expression and neuroprotection in the retina.Mol Neurobiol. 2010 Oct;42(2):124-32. doi: 10.1007/s12035-010-8143-3. Epub 2010 Jun 2. Mol Neurobiol. 2010. PMID: 20514530 Review.
Cited by
-
Decreased ceramide underlies mitochondrial dysfunction in Charcot-Marie-Tooth 2F.FASEB J. 2018 Mar;32(3):1716-1728. doi: 10.1096/fj.201701067R. Epub 2018 Jan 3. FASEB J. 2018. PMID: 29133339 Free PMC article.
-
Mechanical signals activate p38 MAPK pathway-dependent reinforcement of actin via mechanosensitive HspB1.Mol Biol Cell. 2017 Oct 1;28(20):2661-2675. doi: 10.1091/mbc.E17-02-0087. Epub 2017 Aug 2. Mol Biol Cell. 2017. PMID: 28768826 Free PMC article.
-
Noxa/HSP27 complex delays degradation of ubiquitylated IkBα in airway epithelial cells to reduce pulmonary inflammation.Mucosal Immunol. 2018 May;11(3):741-751. doi: 10.1038/mi.2017.117. Epub 2018 Jan 24. Mucosal Immunol. 2018. PMID: 29363670 Free PMC article.
-
Probing the salivary proteome for prognostic biomarkers in response to non-surgical periodontal therapy.J Clin Periodontol. 2025 Jan;52(1):56-67. doi: 10.1111/jcpe.13990. Epub 2024 Apr 25. J Clin Periodontol. 2025. PMID: 38660744 Free PMC article.
-
A keratin scaffold regulates epidermal barrier formation, mitochondrial lipid composition, and activity.J Cell Biol. 2015 Dec 7;211(5):1057-75. doi: 10.1083/jcb.201404147. J Cell Biol. 2015. PMID: 26644517 Free PMC article.
References
-
- Alford KA, Glennie S, Turrell BR, Rawlinson L, Saklatvala J, et al. (2007) Heat shock protein 27 functions in inflammatory gene expression and transforming growth factor-beta-activated kinase-1 (TAK1)-mediated signaling. J Biol Chem 282: 6232–6241. - PubMed
-
- Rousseau S, Houle F, Landry J, Huot J (1997) p38 MAP kinase activation by vascular endothelial growth factor mediates actin reorganization and cell migration in human endothelial cells. Oncogene 15: 2169–2177. - PubMed
-
- Voss OH, Batra S, Kolattukudy SJ, Gonzalez-Mejia ME, Smith JB, et al. (2007) Binding of caspase-3 prodomain to heat shock protein 27 regulates monocyte apoptosis by inhibiting caspase-3 proteolytic activation. J Biol Chem 282: 25088–25099. - PubMed
-
- Jog NR, Jala VR, Ward RA, Rane MJ, Haribabu B, et al. (2007) Heat shock protein 27 regulates neutrophil chemotaxis and exocytosis through two independent mechanisms. J Immunol 178: 2421–2428. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

