Distribution and Ca(2+) signalling of fibroblast-like (PDGFR(+)) cells in the murine gastric fundus
- PMID: 24144881
- PMCID: PMC3892471
- DOI: 10.1113/jphysiol.2013.264747
Distribution and Ca(2+) signalling of fibroblast-like (PDGFR(+)) cells in the murine gastric fundus
Abstract
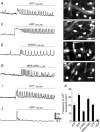
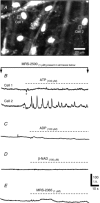
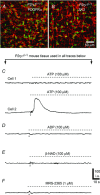
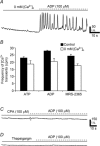
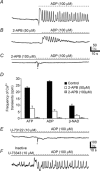
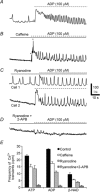
Platelet-derived growth factor receptor α positive (PDGFRα(+)) cells are suggested to mediate purinergic inputs in GI muscles, but the responsiveness of these cells to purines in situ has not been evaluated. We developed techniques to label and visualize PDGFRα(+) cells in murine gastric fundus, load cells with Ca(2+) indicators, and follow their activity via digital imaging. Immunolabelling demonstrated a high density of PDGFRα(+) cells in the fundus. Cells were isolated and purified by fluorescence-activated cell sorting (FACS) using endogenous expression of enhanced green fluorescent protein (eGFP) driven off the Pdgfra promoter. Quantitative PCR showed high levels of expression of purinergic P2Y1 receptors and SK3 K(+) channels in PDGFRα(+) cells. Ca(2+) imaging was used to characterize spontaneous Ca(2+) transients and responses to purines in PDGFRα(+) cells in situ. ATP, ADP, UTP and β-NAD elicited robust Ca(2+) transients in PDGFRα(+) cells. Ca(2+) transients were also elicited by the P2Y1-specific agonist (N)-methanocarba-2MeSADP (MRS-2365), and inhibited by MRS-2500, a P2Y1-specific antagonist. Responses to ADP, MRS-2365 and β-NAD were absent in PDGFRα(+) cells from P2ry1((-/-)) mice, but responses to ATP were retained. Purine-evoked Ca(2+) transients were mediated through Ca(2+) release mechanisms. Inhibitors of phospholipase C (U-73122), IP3 (2-APB), ryanodine receptors (Ryanodine) and SERCA pump (cyclopiazonic acid and thapsigargin) abolished Ca(2+) transients elicited by purines. This study provides a link between purine binding to P2Y1 receptors and activation of SK3 channels in PDGFRα(+) cells. Activation of Ca(2+) release is likely to be the signalling mechanism in PDGFRα(+) cells responsible for the transduction of purinergic enteric inhibitory input in gastric fundus muscles.
Figures






References
-
- Banks BE, Brown C, Burgess GM, Burnstock G, Claret M, Cocks TM, Jenkinson DH. Apamin blocks certain neurotransmitter-induced increases in potassium permeability. Nature. 1979;282:415–417. - PubMed
-
- Bootman MD, Collins TJ, Mackenzie L, Roderick HL, Berridge MJ, Peppiatt CM. 2-Aminoethoxydiphenyl borate (2-APB) is a reliable blocker of store-operated Ca2+ entry but an inconsistent inhibitor of InsP3-induced Ca2+ release. FASEB J. 2002;16:1145–1150. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

