Different involvement of promoter methylation in the expression of organic cation/carnitine transporter 2 (OCTN2) in cancer cell lines
- PMID: 24146874
- PMCID: PMC3797819
- DOI: 10.1371/journal.pone.0076474
Different involvement of promoter methylation in the expression of organic cation/carnitine transporter 2 (OCTN2) in cancer cell lines
Abstract
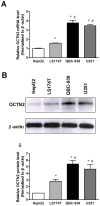
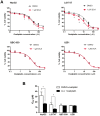
Organic cation/carnitine transporter 2 (OCTN2) is responsible for the cellular uptake of the antineoplastic agent, oxaliplatin. Epigenetic modification is a possible mechanism of altered drug-transporter expression in cancers, leading to altered efficacy of chemotherapeutic drugs. However, the mechanisms governing OCTN2 regulation are not completely understood. In this study, the low levels of OCTN2 in HepG2 and LS174T cells were elevated by the demethylating reagent, decitabine (DCA). To further reveal the epigenetic mechanism of down-regulation of OCTN2, we found that Region-1 within the OCTN2 promoter (spanning -354 to +85) was a determinant of OCTN2 expression in a luciferase reporter assay. Moreover, methylation-specific PCR (MSP) and bisulfite genomic sequencing showed that the degree of individual methylated CpG sites within this region was inversely correlated with the levels of OCTN2 in different cancer cells. Application of DCA to HepG2 and LS174T cells reversed the hypermethylation status of the OCTN2 promoter and increased OCTN2 expression, enhancing cellular uptake of oxaliplatin. Thus, we identified that promoter methylation is responsible for epigenetic down-regulation of OCTN2 in HepG2 and LS174T cells. Given the essential role of OCTN2 in cancer cell uptake of chemotherapeutics, and thus treatment efficacy, pretreatment with a demethylating reagent is a possible strategy for optimizing pharmacotherapies against cancers.
Conflict of interest statement
Figures





Similar articles
-
The Human OCTN Sub-Family: Gene and Protein Structure, Expression, and Regulation.Int J Mol Sci. 2024 Aug 10;25(16):8743. doi: 10.3390/ijms25168743. Int J Mol Sci. 2024. PMID: 39201429 Free PMC article. Review.
-
Luteolin potentiates the sensitivity of colorectal cancer cell lines to oxaliplatin through the PPARγ/OCTN2 pathway.Anticancer Drugs. 2014 Oct;25(9):1016-27. doi: 10.1097/CAD.0000000000000125. Anticancer Drugs. 2014. PMID: 25075794
-
Hypermethylation of growth arrest DNA damage-inducible gene 45 beta promoter in human hepatocellular carcinoma.Am J Pathol. 2004 Nov;165(5):1689-99. doi: 10.1016/s0002-9440(10)63425-6. Am J Pathol. 2004. PMID: 15509538 Free PMC article.
-
Functional genetic variation in the basal promoter of the organic cation/carnitine transporters OCTN1 (SLC22A4) and OCTN2 (SLC22A5).J Pharmacol Exp Ther. 2009 Apr;329(1):262-71. doi: 10.1124/jpet.108.146449. Epub 2009 Jan 13. J Pharmacol Exp Ther. 2009. PMID: 19141711 Free PMC article.
-
OCTN: A Small Transporter Subfamily with Great Relevance to Human Pathophysiology, Drug Discovery, and Diagnostics.SLAS Discov. 2019 Feb;24(2):89-110. doi: 10.1177/2472555218812821. Epub 2018 Dec 7. SLAS Discov. 2019. PMID: 30523710 Review.
Cited by
-
Carnitine Traffic in Cells. Link With Cancer.Front Cell Dev Biol. 2020 Sep 18;8:583850. doi: 10.3389/fcell.2020.583850. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 33072764 Free PMC article. Review.
-
The Human OCTN Sub-Family: Gene and Protein Structure, Expression, and Regulation.Int J Mol Sci. 2024 Aug 10;25(16):8743. doi: 10.3390/ijms25168743. Int J Mol Sci. 2024. PMID: 39201429 Free PMC article. Review.
-
Dysfunction of the carnitine cycle in tumor progression.Heliyon. 2024 Aug 8;10(16):e35961. doi: 10.1016/j.heliyon.2024.e35961. eCollection 2024 Aug 30. Heliyon. 2024. PMID: 39211923 Free PMC article. Review.
-
DNA Methylation-Based Epigenetic Repression of SLC22A4 Promotes Resistance to Cytarabine in Acute Myeloid Leukemia.Clin Transl Sci. 2021 Jan;14(1):137-142. doi: 10.1111/cts.12861. Epub 2020 Sep 9. Clin Transl Sci. 2021. PMID: 32905646 Free PMC article.
-
Inflammation and Organic Cation Transporters Novel (OCTNs).Biomolecules. 2024 Mar 25;14(4):392. doi: 10.3390/biom14040392. Biomolecules. 2024. PMID: 38672410 Free PMC article. Review.
References
-
- Wu X, Prasad PD, Leibach FH, Ganapathy V (1998) cDNA sequence, transport function, and genomic organization of human OCTN2, a new member of the organic cation transporter family. Biochem Biophys Res Commun 246: 589–595. - PubMed
-
- Tamai I, Ohashi R, Nezu J, Yabuuchi H, Oku A, et al. (1998) Molecular and functional identification of sodium ion-dependent, high affinity human carnitine transporter OCTN2. J Biol Chem 273: 20378–20382. - PubMed
-
- Yamazaki K, Takazoe M, Tanaka T, Ichimori T, Saito S, et al. (2004) Association analysis of SLC22A4, SLC22A5 and DLG5 in Japanese patients with Crohn disease. J Hum Genet 49: 664–668. - PubMed
-
- Fisher SA, Hampe J, Onnie CM, Daly MJ, Curley C, et al. (2006) Direct or indirect association in a complex disease: the role of SLC22A4 and SLC22A5 functional variants in Crohn disease. Hum Mutat 27: 778–785. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

