Signal one and two blockade are both critical for non-myeloablative murine HSCT across a major histocompatibility complex barrier
- PMID: 24147041
- PMCID: PMC3798400
- DOI: 10.1371/journal.pone.0077632
Signal one and two blockade are both critical for non-myeloablative murine HSCT across a major histocompatibility complex barrier
Abstract
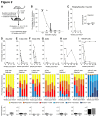
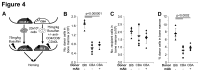
Non-myeloablative allogeneic haematopoietic stem cell transplantation (HSCT) is rarely achievable clinically, except where donor cells have selective advantages. Murine non-myeloablative conditioning regimens have limited clinical success, partly through use of clinically unachievable cell doses or strain combinations permitting allograft acceptance using immunosuppression alone. We found that reducing busulfan conditioning in murine syngeneic HSCT, increases bone marrow (BM):blood SDF-1 ratio and total donor cells homing to BM, but reduces the proportion of donor cells engrafting. Despite this, syngeneic engraftment is achievable with non-myeloablative busulfan (25 mg/kg) and higher cell doses induce increased chimerism. Therefore we investigated regimens promoting initial donor cell engraftment in the major histocompatibility complex barrier mismatched CBA to C57BL/6 allo-transplant model. This requires full myeloablation and immunosuppression with non-depleting anti-CD4/CD8 blocking antibodies to achieve engraftment of low cell doses, and rejects with reduced intensity conditioning (≤75 mg/kg busulfan). We compared increased antibody treatment, G-CSF, niche disruption and high cell dose, using reduced intensity busulfan and CD4/8 blockade in this model. Most treatments increased initial donor engraftment, but only addition of co-stimulatory blockade permitted long-term engraftment with reduced intensity or non-myeloablative conditioning, suggesting that signal 1 and 2 T-cell blockade is more important than early BM niche engraftment for transplant success.
Conflict of interest statement
Figures


References
-
- Wynn RF, Wraith JE, Mercer J, O'Meara A, Tylee K et al. (2009) Improved metabolic correction in patients with lysosomal storage disease treated with hematopoietic stem cell transplant compared with enzyme replacement therapy. J Pediatr 154: 609-611. doi: 10.1016/j.jpeds.2008.11.005. PubMed: 19324223. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

