Endogenous human MDM2-C is highly expressed in human cancers and functions as a p53-independent growth activator
- PMID: 24147044
- PMCID: PMC3795673
- DOI: 10.1371/journal.pone.0077643
Endogenous human MDM2-C is highly expressed in human cancers and functions as a p53-independent growth activator
Abstract
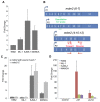
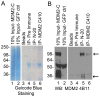
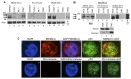
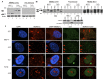
Human cancers over-expressing mdm2, through a T to G variation at a single nucleotide polymorphism at position 309 (mdm2 SNP309), have functionally inactivated p53 that is not effectively degraded. They also have high expression of the alternatively spliced transcript, mdm2-C. Alternatively spliced mdm2 transcripts are expressed in many forms of human cancer and when they are exogenously expressed they transform human cells. However no study to date has detected endogenous MDM2 protein isoforms. Studies with exogenous expression of splice variants have been carried out with mdm2-A and mdm2-B, but the mdm2-C isoform has remained virtually unexplored. We addressed the cellular influence of exogenously expressed MDM2-C, and asked if endogenous MDM2-C protein was present in human cancers. To detect endogenous MDM2-C protein, we created a human MDM2-C antibody to the splice junction epitope of exons four and ten (MDM2 C410) and validated the antibody with in vitro translated full length MDM2 compared to MDM2-C. Interestingly, we discovered that MDM2-C co-migrates with MDM2-FL at approximately 98 kDa. Using the validated C410 antibody, we detected high expression of endogenous MDM2-C in human cancer cell lines and human cancer tissues. In the estrogen receptor positive (ER+) mdm2 G/G SNP309 breast cancer cell line, T47D, we observed an increase in endogenous MDM2-C protein with estrogen treatment. MDM2-C localized to the nucleus and the cytoplasm. We examined the biological activity of MDM2-C by exogenously expressing the protein and observed that MDM2-C did not efficiently target p53 for degradation or reduce p53 transcriptional activity. Exogenous expression of MDM2-C in p53-null human cancer cells increased colony formation, indicating p53-independent tumorigenic properties. Our data indicate a role for MDM2-C that does not require the inhibition of p53 for increasing cancer cell proliferation and survival.
Conflict of interest statement
Figures



Similar articles
-
Disruption of the p53-Mdm2 complex by Nutlin-3 reveals different cancer cell phenotypes.Ethn Dis. 2008 Spring;18(2 Suppl 2):S2-1-8. Ethn Dis. 2008. PMID: 18646312 Free PMC article.
-
Homozygous mdm2 SNP309 cancer cells with compromised transcriptional elongation at p53 target genes are sensitive to induction of p53-independent cell death.Oncotarget. 2015 Oct 27;6(33):34573-91. doi: 10.18632/oncotarget.5312. Oncotarget. 2015. PMID: 26416444 Free PMC article.
-
Stress-induced alternative splice forms of MDM2 and MDMX modulate the p53-pathway in distinct ways.PLoS One. 2014 Aug 8;9(8):e104444. doi: 10.1371/journal.pone.0104444. eCollection 2014. PLoS One. 2014. PMID: 25105592 Free PMC article.
-
The regulation of MDM2 oncogene and its impact on human cancers.Acta Biochim Biophys Sin (Shanghai). 2014 Mar;46(3):180-9. doi: 10.1093/abbs/gmt147. Epub 2014 Jan 3. Acta Biochim Biophys Sin (Shanghai). 2014. PMID: 24389645 Free PMC article. Review.
-
p53 isoforms - a conspiracy to kidnap p53 tumor suppressor activity?Cell Mol Life Sci. 2009 Feb;66(3):391-406. doi: 10.1007/s00018-008-8336-3. Cell Mol Life Sci. 2009. PMID: 18854945 Free PMC article. Review.
Cited by
-
MDM2, MDM2-C, and mutant p53 expression influence breast cancer survival in a multiethnic population.Breast Cancer Res Treat. 2019 Feb;174(1):257-269. doi: 10.1007/s10549-018-5065-7. Epub 2018 Nov 23. Breast Cancer Res Treat. 2019. PMID: 30470976 Free PMC article.
-
Oncogenic Functions of Alternatively Spliced MDM2-ALT2 Isoform in Retroperitoneal Liposarcoma.Int J Mol Sci. 2024 Dec 17;25(24):13516. doi: 10.3390/ijms252413516. Int J Mol Sci. 2024. PMID: 39769278 Free PMC article.
-
Mutant p53 in Cancer: Accumulation, Gain-of-Function, and Therapy.J Mol Biol. 2017 Jun 2;429(11):1595-1606. doi: 10.1016/j.jmb.2017.03.030. Epub 2017 Apr 6. J Mol Biol. 2017. PMID: 28390900 Free PMC article. Review.
-
MDM2-C Functions as an E3 Ubiquitin Ligase.Cancer Manag Res. 2020 Aug 24;12:7715-7724. doi: 10.2147/CMAR.S260943. eCollection 2020. Cancer Manag Res. 2020. PMID: 32904724 Free PMC article.
-
The Roles of MDM2 and MDMX in Cancer.Annu Rev Pathol. 2016 May 23;11:617-44. doi: 10.1146/annurev-pathol-012414-040349. Epub 2016 Mar 17. Annu Rev Pathol. 2016. PMID: 27022975 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

