Effect of temperature on crossbridge force changes during fatigue and recovery in intact mouse muscle fibers
- PMID: 24147145
- PMCID: PMC3798468
- DOI: 10.1371/journal.pone.0078918
Effect of temperature on crossbridge force changes during fatigue and recovery in intact mouse muscle fibers
Abstract
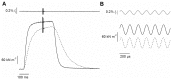
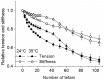
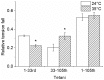
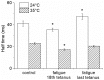
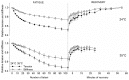
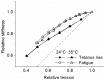
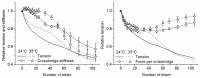
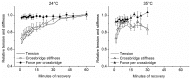
Repetitive or prolonged muscle contractions induce muscular fatigue, defined as the inability of the muscle to maintain the initial tension or power output. In the present experiments, made on intact fiber bundles from FDB mouse, fatigue and recovery from fatigue were investigated at 24°C and 35°C. Force and stiffness were measured during tetani elicited every 90 s during the pre-fatigue control phase and recovery and every 1.5 s during the fatiguing phase made of 105 consecutive tetani. The results showed that force decline could be split in an initial phase followed by a later one. Loss of force during the first phase was smaller and slower at 35°C than at 24°C, whereas force decline during the later phase was greater at 35°C so that total force depression at the end of fatigue was the same at both temperatures. The initial force decline occurred without great reduction of fiber stiffness and was attributed to a decrease of the average force per attached crossbridge. Force decline during the later phase was accompanied by a proportional stiffness decrease and was attributed to a decrease of the number of attached crossbridge. Similarly to fatigue, at both 24 and 35°C, force recovery occurred in two phases: the first associated with the recovery of the average force per attached crossbridge and the second due to the recovery of the pre-fatigue attached crossbridge number. These changes, symmetrical to those occurring during fatigue, are consistent with the idea that, i) initial phase is due to the direct fast inhibitory effect of [Pi]i increase during fatigue on crossbridge force; ii) the second phase is due to the delayed reduction of Ca(2+) release and /or reduction of the Ca(2+) sensitivity of the myofibrils due to high [Pi]i.
Conflict of interest statement
Figures
Similar articles
-
Phosphate increase during fatigue affects crossbridge kinetics in intact mouse muscle at physiological temperature.J Physiol. 2017 Jul 1;595(13):4317-4328. doi: 10.1113/JP273672. Epub 2017 May 8. J Physiol. 2017. PMID: 28332714 Free PMC article.
-
Changes in crossbridge and non-crossbridge energetics during moderate fatigue of frog muscle fibres.J Physiol. 1993 Aug;468:543-56. doi: 10.1113/jphysiol.1993.sp019787. J Physiol. 1993. PMID: 8254523 Free PMC article.
-
Changes of the force-velocity relation, isometric tension and relaxation rate during fatigue in intact, single fibres of Xenopus skeletal muscle.J Muscle Res Cell Motil. 1994 Jun;15(3):287-98. doi: 10.1007/BF00123481. J Muscle Res Cell Motil. 1994. PMID: 7929794
-
Effects of Hydrostatic-Pressure on Muscle Contraction: A Look Back on Some Experimental Findings.Int J Mol Sci. 2023 Mar 6;24(5):5031. doi: 10.3390/ijms24055031. Int J Mol Sci. 2023. PMID: 36902460 Free PMC article. Review.
-
Muscle contraction and fatigue. The role of adenosine 5'-diphosphate and inorganic phosphate.Sports Med. 1997 May;23(5):287-305. doi: 10.2165/00007256-199723050-00003. Sports Med. 1997. PMID: 9181667 Review.
Cited by
-
Mechanical isolation, and measurement of force and myoplasmic free [Ca2+] in fully intact single skeletal muscle fibers.Nat Protoc. 2017 Sep;12(9):1763-1776. doi: 10.1038/nprot.2017.056. Epub 2017 Aug 3. Nat Protoc. 2017. PMID: 28771237
-
The effects of fatigue and oxidation on contractile function of intact muscle fibers and myofibrils isolated from the mouse diaphragm.Sci Rep. 2019 Mar 14;9(1):4422. doi: 10.1038/s41598-019-39353-5. Sci Rep. 2019. PMID: 30872655 Free PMC article.
-
Effects of knee flexor submaximal isometric contraction until exhaustion on semitendinosus and biceps femoris long head shear modulus in healthy individuals.Sci Rep. 2020 Oct 2;10(1):16433. doi: 10.1038/s41598-020-73433-1. Sci Rep. 2020. PMID: 33009453 Free PMC article.
-
The Effects of Isometric Fatigue on Trunk Muscle Stiffness: Implications for Shear-Wave Elastography Measurements.Sensors (Basel). 2022 Dec 4;22(23):9476. doi: 10.3390/s22239476. Sensors (Basel). 2022. PMID: 36502176 Free PMC article.
-
The likely effects of thermal climate change on vertebrate skeletal muscle mechanics with possible consequences for animal movement and behaviour.Conserv Physiol. 2019 Oct 31;7(1):coz066. doi: 10.1093/conphys/coz066. eCollection 2019. Conserv Physiol. 2019. PMID: 31687144 Free PMC article. Review.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

