Anticancer activity of small-molecule and nanoparticulate arsenic(III) complexes
- PMID: 24147771
- PMCID: PMC3893798
- DOI: 10.1021/ic401211u
Anticancer activity of small-molecule and nanoparticulate arsenic(III) complexes
Abstract
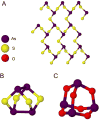
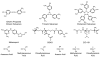
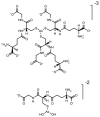
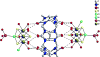
Starting in ancient China and Greece, arsenic-containing compounds have been used in the treatment of disease for over 3000 years. They were used for a variety of diseases in the 20th century, including parasitic and sexually transmitted illnesses. A resurgence of interest in the therapeutic application of arsenicals has been driven by the discovery that low doses of a 1% aqueous solution of arsenic trioxide (i.e., arsenous acid) lead to complete remission of certain types of leukemia. Since Food and Drug Administration (FDA) approval of arsenic trioxide (As2O3) for treatment of acute promyelocytic leukemia in 2000, it has become a front-line therapy in this indication. There are currently over 100 active clinical trials involving inorganic arsenic or organoarsenic compounds registered with the FDA for the treatment of cancers. New generations of inorganic and organometallic arsenic compounds with enhanced activity or targeted cytotoxicity are being developed to overcome some of the shortcomings of arsenic therapeutics, namely, short plasma half-lives and a narrow therapeutic window.
Figures

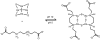


Similar articles
-
Arsenic trioxide and acute promyelocytic leukemia: clinical and biological.Curr Top Microbiol Immunol. 2007;313:129-44. doi: 10.1007/978-3-540-34594-7_8. Curr Top Microbiol Immunol. 2007. PMID: 17217042 Review.
-
Treatment of acute promyelocytic leukemia with arsenic trioxide: clinical and basic studies.Leuk Lymphoma. 2001 Nov-Dec;42(6):1265-73. doi: 10.3109/10428190109097751. Leuk Lymphoma. 2001. PMID: 11911407 Review.
-
Use of arsenic trioxide (As2O3) in the treatment of acute promyelocytic leukemia (APL): II. Clinical efficacy and pharmacokinetics in relapsed patients.Blood. 1997 May 1;89(9):3354-60. Blood. 1997. PMID: 9129042 Clinical Trial.
-
Arsenic trioxide.Expert Opin Pharmacother. 2008 Jul;9(10):1773-85. doi: 10.1517/14656566.9.10.1773. Expert Opin Pharmacother. 2008. PMID: 18570609 Review.
-
Arsenic trioxide as first-line treatment for acute promyelocytic leukemia.Am J Health Syst Pharm. 2009 Nov 1;66(21):1913-8. doi: 10.2146/ajhp080342. Am J Health Syst Pharm. 2009. PMID: 19850784
Cited by
-
Convertible MRI contrast: Sensing the delivery and release of anti-glioma nano-drugs.Sci Rep. 2015 May 12;5:9874. doi: 10.1038/srep09874. Sci Rep. 2015. PMID: 25962872 Free PMC article.
-
Erythrocyte Membrane-Coated Arsenic Trioxide-Loaded Sodium Alginate Nanoparticles for Tumor Therapy.Pharmaceutics. 2019 Dec 24;12(1):21. doi: 10.3390/pharmaceutics12010021. Pharmaceutics. 2019. PMID: 31878155 Free PMC article.
-
Organoarsenic Compounds with In Vitro Activity against the Malaria Parasite Plasmodium falciparum.Biomedicines. 2020 Aug 2;8(8):260. doi: 10.3390/biomedicines8080260. Biomedicines. 2020. PMID: 32748808 Free PMC article.
-
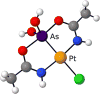
Crystal structures of chlorido-[dihy-droxybis-(1-imino-eth-oxy)]arsanido-κ3 N,As,N']platinum(II) and of a polymorph of chlorido-[dihy-droxybis-(1-imino-prop-oxy)arsanido-κ3 N,As,N']platinum(II).Acta Crystallogr E Crystallogr Commun. 2020 Jan 10;76(Pt 2):180-185. doi: 10.1107/S2056989019016463. eCollection 2020 Feb 1. Acta Crystallogr E Crystallogr Commun. 2020. PMID: 32071743 Free PMC article.
-
Radiochemistry: A Hot Field with Opportunities for Cool Chemistry.ACS Cent Sci. 2023 Nov 14;9(12):2183-2195. doi: 10.1021/acscentsci.3c01050. eCollection 2023 Dec 27. ACS Cent Sci. 2023. PMID: 38161375 Free PMC article. Review.
References
-
- Parascandola J. King of Poisons: A History of Arsenic. 2012:134–134.
-
- Konkola K. J Hist Med Allied Sci. 1992;47:186–209. - PubMed
-
- Przygoda G, Feldmann Jr, Cullen WR. Appl Organomet Chem. 2001;15:457–462.
-
- Haller JS. Pharmacy in History. 1975;17:87–100. - PubMed
-
- Lu DP, Wang Q. Int J Hematol. 2002;76(Suppl 1):316–8. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

