The antioxidant effects of the flavonoids rutin and quercetin inhibit oxaliplatin-induced chronic painful peripheral neuropathy
- PMID: 24152430
- PMCID: PMC3835704
- DOI: 10.1186/1744-8069-9-53
The antioxidant effects of the flavonoids rutin and quercetin inhibit oxaliplatin-induced chronic painful peripheral neuropathy
Abstract
Background: Oxaliplatin, the third-generation platinum compound, has evolved as one of the most important therapeutic agents in colorectal cancer chemotherapy. The main limiting factor in oxaliplatin treatment is painful neuropathy that is difficult to treat. This side effect has been studied for several years, but its full mechanism is still inconclusive, and effective treatment does not exist. Data suggest that oxaliplatin's initial neurotoxic effect is peripheral and oxidative stress-dependent. A spinal target is also suggested in its mechanism of action. The flavonoids rutin and quercetin have been described as cell-protecting agents because of their antioxidant, antinociceptive, and anti-inflammatory actions. We proposed a preventive effect of these agents on oxaliplatin-induced painful peripheral neuropathy based on their antioxidant properties.
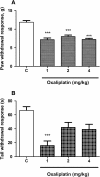
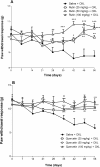
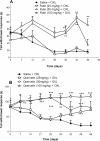
Methods: Oxaliplatin (1 mg/kg, i.v.) was injected in male Swiss mice, twice a week (total of nine injections). The development of sensory alterations, such as thermal and mechanical allodynia, was evaluated using the tail immersion test in cold water (10°C) and the von Frey test. Rutin and quercetin (25-100 mg/kg, i.p.) were injected 30 min before each oxaliplatin injection. The animals' spinal cords were removed for histopathological and immunohistochemical evaluation and malondialdehyde assay.
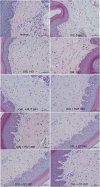
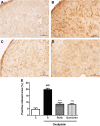
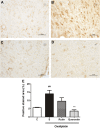
Results: Oxaliplatin significantly increased thermal and mechanical nociceptive response, effects prevented by quercetin and rutin at all doses. Fos immunostaining in the dorsal horn of the spinal cord confirmed these results. The oxidative stress assays mainly showed that oxaliplatin induced peroxidation in the spinal cord and that rutin and quercetin decreased this effect. The flavonoids also decreased inducible nitric oxide synthase and nitrotyrosine immunostaining in the dorsal horn of the spinal cord. These results suggest that nitric oxide and peroxynitrite are also involved in the neurotoxic effect of oxaliplatin and that rutin and quercetin can inhibit their effect in the spinal cord. We also observed the preservation of dorsal horn structure using histopathological analyses.
Conclusions: Oxaliplatin induced painful peripheral neuropathy in mice, an effect that was prevented by rutin and quercetin. The mechanism of action of oxaliplatin appears to be, at least, partially oxidative stress-induced damage in dorsal horn neurons, with the involvement of lipid peroxidation and protein nitrosylation.
Figures



References
-
- Chu E. In: The Clinical Use of Oxaliplatin: Case Studies and Roundtable Discussion. Marshall JL, editor. Manhasset: Oncology Pub Division of CMP Healthcare Media; 2004. The clinical Use of Oxaliplatin: The new member of the class; p. 109.
-
- De Gramont A, Figer A, Seymour M, Homerin M, Hmissi A, Cassidy J, Boni C, Cortes-Funes H, Cervantes A, Freyer G, Papamichael D, Le Bail N, Louvet C, Hendler D, de Brand F, Wilson C, Morvan F, Bonetti A. Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol. 2000;18:2938–2947. - PubMed
-
- Goldberg RM, Sargent DJ, Morton RF, Fuchs CS, Ramanathan RK, Williamson SK, Findlay BP, Pitot HC, Alberts SR. A randomized controlled trial of fluorouracil plus leucovorin, irinotecan, and oxaliplatin combinations in patients with previously untreated metastatic colorectal cancer. J Clin Oncol. 2003;22:23–30. doi: 10.1200/JCO.2004.09.046. - DOI - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

