Human plasmacytoid dendritic cells sense lymphocytic choriomeningitis virus-infected cells in vitro
- PMID: 24155390
- PMCID: PMC3911694
- DOI: 10.1128/JVI.01714-13
Human plasmacytoid dendritic cells sense lymphocytic choriomeningitis virus-infected cells in vitro
Abstract
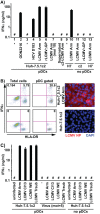
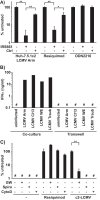
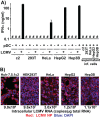
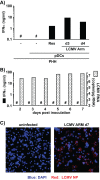
We previously reported that exosomal transfer of hepatitis C virus (HCV) positive-strand RNA from human Huh-7 hepatoma cells to human plasmacytoid dendritic cells (pDCs) triggers pDC alpha/beta interferon (IFN-α/β) production in a Toll-like receptor 7 (TLR7)-dependent, virus-independent manner. Here we show that human pDCs are also activated by a TLR7-dependent, virus-independent, exosomal RNA transfer mechanism by human and mouse hepatoma and nonhepatoma cells that replicate the negative-strand lymphocytic choriomeningitis virus (LCMV).
Figures
Similar articles
-
Resistance of human plasmacytoid dendritic CAL-1 cells to infection with lymphocytic choriomeningitis virus (LCMV) is caused by restricted virus cell entry, which is overcome by contact of CAL-1 cells with LCMV-infected cells.Virology. 2017 Nov;511:106-113. doi: 10.1016/j.virol.2017.08.017. Epub 2017 Aug 24. Virology. 2017. PMID: 28843812 Free PMC article.
-
Interleukin-27R Signaling Mediates Early Viral Containment and Impacts Innate and Adaptive Immunity after Chronic Lymphocytic Choriomeningitis Virus Infection.J Virol. 2018 May 29;92(12):e02196-17. doi: 10.1128/JVI.02196-17. Print 2018 Jun 15. J Virol. 2018. PMID: 29593047 Free PMC article.
-
Plasmacytoid dendritic cells sense hepatitis C virus-infected cells, produce interferon, and inhibit infection.Proc Natl Acad Sci U S A. 2010 Apr 20;107(16):7431-6. doi: 10.1073/pnas.1002301107. Epub 2010 Mar 15. Proc Natl Acad Sci U S A. 2010. PMID: 20231459 Free PMC article.
-
Infection of dendritic cells by lymphocytic choriomeningitis virus.Curr Top Microbiol Immunol. 2003;276:125-44. doi: 10.1007/978-3-662-06508-2_6. Curr Top Microbiol Immunol. 2003. PMID: 12797446 Free PMC article. Review.
-
Regulation of TLR7/9 signaling in plasmacytoid dendritic cells.Protein Cell. 2013 Jan;4(1):40-52. doi: 10.1007/s13238-012-2104-8. Epub 2012 Nov 7. Protein Cell. 2013. PMID: 23132256 Free PMC article. Review.
Cited by
-
Sensing of Porcine Reproductive and Respiratory Syndrome Virus-Infected Macrophages by Plasmacytoid Dendritic Cells.Front Microbiol. 2016 Jun 2;7:771. doi: 10.3389/fmicb.2016.00771. eCollection 2016. Front Microbiol. 2016. PMID: 27458429 Free PMC article.
-
Respiratory Syncytial Virus Infection Changes Cargo Composition of Exosome Released from Airway Epithelial Cells.Sci Rep. 2018 Jan 10;8(1):387. doi: 10.1038/s41598-017-18672-5. Sci Rep. 2018. PMID: 29321591 Free PMC article.
-
Regulation of the Host Antiviral State by Intercellular Communications.Viruses. 2015 Aug 19;7(8):4707-33. doi: 10.3390/v7082840. Viruses. 2015. PMID: 26295405 Free PMC article. Review.
-
STING-Licensed Macrophages Prime Type I IFN Production by Plasmacytoid Dendritic Cells in the Bone Marrow during Severe Plasmodium yoelii Malaria.PLoS Pathog. 2016 Oct 28;12(10):e1005975. doi: 10.1371/journal.ppat.1005975. eCollection 2016 Oct. PLoS Pathog. 2016. PMID: 27792766 Free PMC article.
-
Exosomes originating from infection with the cytoplasmic single-stranded RNA virus Rift Valley fever virus (RVFV) protect recipient cells by inducing RIG-I mediated IFN-B response that leads to activation of autophagy.Cell Biosci. 2021 Dec 25;11(1):220. doi: 10.1186/s13578-021-00732-z. Cell Biosci. 2021. PMID: 34953502 Free PMC article.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

