Metagenomic approach for the isolation of a thermostable β-galactosidase with high tolerance of galactose and glucose from soil samples of Turpan Basin
- PMID: 24156692
- PMCID: PMC4016535
- DOI: 10.1186/1471-2180-13-237
Metagenomic approach for the isolation of a thermostable β-galactosidase with high tolerance of galactose and glucose from soil samples of Turpan Basin
Abstract
Background: β-Galactosidases can be used to produce low-lactose milk and dairy products for lactose intolerant people. Although commercial β-galactosidases have outstanding lactose hydrolysis ability, their thermostability is low, and reaction products have strong inhibition to these enzymes. In addition, the β-galactosidases possessing simultaneously high thermostability and tolerance of galactose and glucose are still seldom reported until now. Therefore, identification of novel β-galactosidases with high thermostability and tolerance to reaction products from unculturable microorganisms accounting for over 99% of microorganisms in the environment via metagenomic strategy is still urgently in demand.
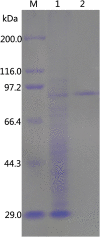
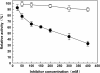
Results: In the present study, a novel β-galactosidase (Gal308) consisting of 658 amino acids was identified from a metagenomic library from soil samples of Turpan Basin in China by functional screening. After being overexpressed in Escherichia coli and purified to homogeneity, the enzymatic properties of Gal308 with N-terminal fusion tag were investigated. The recombinant enzyme displayed a pH optimum of 6.8 and a temperature optimum of 78 °C, and was considerably stable in the temperature range of 40 °C - 70 °C with almost unchangeable activity after incubation for 60 min. Furthermore, Gal308 displayed a very high tolerance of galactose and glucose, with the highest inhibition constant K(i,gal) (238 mM) and K(i,glu) (1725 mM) among β-galactosidases. In addition, Gal308 also exhibited high enzymatic activity for its synthetic substrate o-nitrophenyl-β-D-galactopyranoside (ONPG, 185 U/mg) and natural substrate lactose (47.6 U/mg).
Conclusion: This study will enrich the source of β-galactosidases, and attract some attentions to β-galactosidases from extreme habitats and metagenomic library. Furthermore, the recombinant Gal308 fused with 156 amino acids exhibits many novel properties including high activity and thermostability at high temperatures, the pH optimum of 6.8, high enzyme activity for lactose, as well as high tolerance of galactose and glucose. These properties make it a good candidate in the production of low-lactose milk and dairy products after further study.
Figures

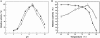
Similar articles
-
A novel cold-active β-D-galactosidase from the Paracoccus sp. 32d--gene cloning, purification and characterization.Microb Cell Fact. 2011 Dec 13;10:108. doi: 10.1186/1475-2859-10-108. Microb Cell Fact. 2011. PMID: 22166118 Free PMC article.
-
Effects of galactose and glucose on the hydrolysis reaction of a thermostable beta-galactosidase from Caldicellulosiruptor saccharolyticus.Appl Microbiol Biotechnol. 2010 Feb;85(5):1427-35. doi: 10.1007/s00253-009-2165-7. Epub 2009 Aug 7. Appl Microbiol Biotechnol. 2010. PMID: 19662397
-
Novel high-performance metagenome β-galactosidases for lactose hydrolysis in the dairy industry.J Biotechnol. 2015 Sep 20;210:27-37. doi: 10.1016/j.jbiotec.2015.06.411. Epub 2015 Jun 26. J Biotechnol. 2015. PMID: 26122513
-
Heat-Loving β-Galactosidases from Cultured and Uncultured Microorganisms.Curr Protein Pept Sci. 2018;19(12):1224-1234. doi: 10.2174/1389203719666180809111659. Curr Protein Pept Sci. 2018. PMID: 30091411 Review.
-
β-Galactosidase: a traditional enzyme given multiple roles through protein engineering.Crit Rev Food Sci Nutr. 2025;65(7):1306-1325. doi: 10.1080/10408398.2023.2292282. Epub 2023 Dec 18. Crit Rev Food Sci Nutr. 2025. PMID: 38108277 Review.
Cited by
-
Metagenomics of Thermophiles with a Focus on Discovery of Novel Thermozymes.Front Microbiol. 2016 Sep 27;7:1521. doi: 10.3389/fmicb.2016.01521. eCollection 2016. Front Microbiol. 2016. PMID: 27729905 Free PMC article. Review.
-
Site-directed mutation of β-galactosidase from Aspergillus candidus to reduce galactose inhibition in lactose hydrolysis.3 Biotech. 2018 Nov;8(11):452. doi: 10.1007/s13205-018-1418-5. Epub 2018 Oct 16. 3 Biotech. 2018. PMID: 30333954 Free PMC article.
-
Core-shell fibremats comprising a poly(AM/DAAM)/ADH nanofibre core and nylon6 shell layer are an attractive immobilization platform for constructing immobilised enzymes.RSC Adv. 2022 Dec 7;12(54):34931-34940. doi: 10.1039/d2ra06620c. eCollection 2022 Dec 6. RSC Adv. 2022. PMID: 36540265 Free PMC article.
-
β-Galactosidases from a Sequence-Based Metagenome: Cloning, Expression, Purification and Characterization.Microorganisms. 2020 Dec 28;9(1):55. doi: 10.3390/microorganisms9010055. Microorganisms. 2020. PMID: 33379234 Free PMC article.
-
Prokaryotic taxonomy and functional diversity assessment of different sequencing platform in a hyper-arid Gobi soil in Xinjiang Turpan Basin, China.Front Microbiol. 2023 Nov 14;14:1211915. doi: 10.3389/fmicb.2023.1211915. eCollection 2023. Front Microbiol. 2023. PMID: 38033567 Free PMC article.
References
-
- Neri DFM, Balcao VM, Carneiro-da-Cunha MG, Carvalho LB Jr, Teixeira JA. Immobilization of β-galactosidase from Kluyveromyces lactis onto a polysiloxane-polyvinyl alcohol magnetic (mPOS-PVA) composite for lactose hydrolysis. Catal Comm. 2008;13:234–239.
-
- Aehle W. Enzymes in industry: production and applications. 2. Weinheim: Wiley-VCH; 2004.
-
- Oliveira C, Guimarães PMR, Domingues L. Recombinant microbial systems for improved β-galactosidase production and biotechnological applications. Biotechnol Adv. 2011;13:600–609. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

