Cloning and expression analysis of a novel high-mobility group box 2 homologue from Lampetra japonica
- PMID: 24158500
- PMCID: PMC3948571
- DOI: 10.1007/s10695-013-9871-x
Cloning and expression analysis of a novel high-mobility group box 2 homologue from Lampetra japonica
Abstract
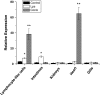
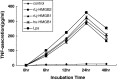
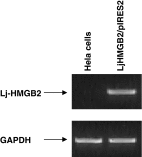
High-mobility group box 2 (HMGB2) is a nonhistone architectural protein that plays important roles in many biological processes. In this study, we cloned a homologue of the HMGB2 from the lymphocyte-like cells of Lampetra japonica (L. japonica). Sequence analysis reveals that L. japonica HMGB2 contains two highly conserved motifs and shares more than 70 % identity with the homologues from other vertebrate species. Subsequently, Lj-HMGB2 was subcloned into the pET-28a(+) and pIRES2 AcGFP1-Nuc vector and expressed in Rosetta blue (DE3) and Hela cell lines, respectively. The recombinant L. japonica HMGB2 (rLj-HMGB2) with apparent molecular mass of 22 kDa was further purified by His-Bind affinity chromatography. Real-time quantitative PCR indicates that the expression level of Lj-HMGB2 was particularly up-regulated in intestines after challenged with lipopolysaccharide, while up-regulated in lymphocyte-like cells and heart after challenged with concanavalin A in vivo. In addition, rLj-HMGB2 could induce the generation of proinflammatory mediators in the activated human acute monocytic leukemia cell line (THP1), which suggested that Lj-HMGB2 may participate in the immune response of the lampreys.
Figures



Similar articles
-
Molecular cloning, expression and antioxidant activity of a peroxiredoxin 2 homologue from Lampetra japonica.Fish Shellfish Immunol. 2010 May-Jun;28(5-6):795-801. doi: 10.1016/j.fsi.2010.01.018. Epub 2010 Feb 6. Fish Shellfish Immunol. 2010. PMID: 20139002
-
Characterizations of two grass carp Ctenopharyngodon idella HMGB2 genes and potential roles in innate immunity.Dev Comp Immunol. 2013 Oct;41(2):164-77. doi: 10.1016/j.dci.2013.06.002. Epub 2013 Jun 10. Dev Comp Immunol. 2013. PMID: 23756189
-
Novel neutrophil inhibitory factor homologue in the buccal gland secretion of Lampetra japonica.Biol Chem. 2011 Jul;392(7):609-16. doi: 10.1515/BC.2011.065. Epub 2011 Jun 1. Biol Chem. 2011. PMID: 21627537
-
Identification and characterization of the lamprey high-mobility group box 1 gene.PLoS One. 2012;7(4):e35755. doi: 10.1371/journal.pone.0035755. Epub 2012 Apr 26. PLoS One. 2012. PMID: 22563397 Free PMC article.
-
HMGB proteins and arthritis.Hum Cell. 2018 Jan;31(1):1-9. doi: 10.1007/s13577-017-0182-x. Epub 2017 Sep 15. Hum Cell. 2018. PMID: 28916968 Free PMC article. Review.
References
-
- Boonyaratanakornkit V, Melvin V, Prendergast P, Altmann M, Ronfani L, Bianchi ME, Taraseviciene L, Nordeen SK, Allegretto EA, Edwards DP. High-mobility group chromatin proteins 1 and 2 functionally interact with steroid hormone receptors to enhance their DNA binding in vitro and transcriptional activity in mammalian cells. Mol Cell Biol. 1998;18:4471–4487. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

