A microchip for quantitative analysis of CNS axon growth under localized biomolecular treatments
- PMID: 24161788
- PMCID: PMC3863683
- DOI: 10.1016/j.jneumeth.2013.09.018
A microchip for quantitative analysis of CNS axon growth under localized biomolecular treatments
Abstract
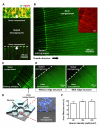
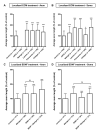
Growth capability of neurons is an essential factor in axon regeneration. To better understand how microenvironments influence axon growth, methods that allow spatial control of cellular microenvironments and easy quantification of axon growth are critically needed. Here, we present a microchip capable of physically guiding the growth directions of axons while providing physical and fluidic isolation from neuronal somata/dendrites that enables localized biomolecular treatments and linear axon growth. The microchip allows axons to grow in straight lines inside the axon compartments even after the isolation; therefore, significantly facilitating the axon length quantification process. We further developed an image processing algorithm that automatically quantifies axon growth. The effect of localized extracellular matrix components and brain-derived neurotropic factor treatments on axon growth was investigated. Results show that biomolecules may have substantially different effects on axon growth depending on where they act. For example, while chondroitin sulfate proteoglycan causes axon retraction when added to the axons, it promotes axon growth when applied to the somata. The newly developed microchip overcomes limitations of conventional axon growth research methods that lack localized control of biomolecular environments and are often performed at a significantly lower cell density for only a short period of time due to difficulty in monitoring of axonal growth. This microchip may serve as a powerful tool for investigating factors that promote axon growth and regeneration.
Keywords: Automated measurement; Axon growth rate; Compartmentalized culture; Localized biomolecular treatment; Microfluidic culture platform; Quantitative axon length analysis.
Copyright © 2013 Elsevier B.V. All rights reserved.
Figures




Similar articles
-
Multi-compartment neuron-glia co-culture platform for localized CNS axon-glia interaction study.Lab Chip. 2012 Sep 21;12(18):3296-304. doi: 10.1039/c2lc40303j. Epub 2012 Jul 24. Lab Chip. 2012. PMID: 22828584 Free PMC article.
-
Axon length quantification microfluidic culture platform for growth and regeneration study.Methods Mol Biol. 2014;1162:85-95. doi: 10.1007/978-1-4939-0777-9_7. Methods Mol Biol. 2014. PMID: 24838960 Free PMC article.
-
A Microchip for High-throughput Axon Growth Drug Screening.Micromachines (Basel). 2016 Jul;7(7):114. doi: 10.3390/mi7070114. Epub 2016 Jul 7. Micromachines (Basel). 2016. PMID: 27928514 Free PMC article.
-
The role of neurotrophic factors in nerve regeneration.Neurosurg Focus. 2009 Feb;26(2):E3. doi: 10.3171/FOC.2009.26.2.E3. Neurosurg Focus. 2009. PMID: 19228105 Review.
-
Spinal cord repair: strategies to promote axon regeneration.Neurobiol Dis. 2001 Feb;8(1):11-8. doi: 10.1006/nbdi.2000.0359. Neurobiol Dis. 2001. PMID: 11162236 Review.
Cited by
-
Recent progress in translational engineered in vitro models of the central nervous system.Brain. 2020 Dec 5;143(11):3181-3213. doi: 10.1093/brain/awaa268. Brain. 2020. PMID: 33020798 Free PMC article. Review.
-
Microfluidic platforms for single neuron analysis.Mater Today Bio. 2022 Feb 16;13:100222. doi: 10.1016/j.mtbio.2022.100222. eCollection 2022 Jan. Mater Today Bio. 2022. PMID: 35243297 Free PMC article. Review.
-
Signal Propagation between Neuronal Populations Controlled by Micropatterning.Front Bioeng Biotechnol. 2016 Jun 15;4:46. doi: 10.3389/fbioe.2016.00046. eCollection 2016. Front Bioeng Biotechnol. 2016. PMID: 27379230 Free PMC article.
-
Nanogrooves for 2D and 3D Microenvironments of SH-SY5Y Cultures in Brain-on-Chip Technology.Front Neurosci. 2020 Jun 24;14:666. doi: 10.3389/fnins.2020.00666. eCollection 2020. Front Neurosci. 2020. PMID: 32670014 Free PMC article.
-
A magnetically actuated microrobot for targeted neural cell delivery and selective connection of neural networks.Sci Adv. 2020 Sep 25;6(39):eabb5696. doi: 10.1126/sciadv.abb5696. Print 2020 Sep. Sci Adv. 2020. PMID: 32978164 Free PMC article.
References
-
- Acheson A, Conover JC, Fandl JP, DeChiara TM, Russell M, Thadani A, Squinto SP, Yancopoulos GD, Lindsay RM. A BDNF autocrine loop in adult sensory neurons prevents cell death. Nature. 1995;374:450–3. - PubMed
-
- Baron-Van Evercooren A, Kleinman HK, Ohno S, Marangos P, Schwartz JP, Dubois-Dalcq ME. Nerve growth factor, laminin, and fibronectin promote neurite growth in human fetal sensory ganglia cultures. Journal of Neuroscience Research. 1982;8:179–93. - PubMed
-
- Benowitz LI, Goldberg DE, Irwin N. Inosine stimulates axon growth in vitro and in the adult CNS. Prog. Brain Res. 2002;137:389–99. - PubMed
-
- Brewer G, Torricelli J, Evege E, Price P. Optimized survival of hippocampal neurons in B27 B27-supplemented neurobasal™, a new serum-free medium combination. Journal of neuroscience research. 1993;35:567–76. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

