Chronic follicular bronchiolitis requires antigen-specific regulatory T cell control to prevent fatal disease progression
- PMID: 24163409
- PMCID: PMC3893829
- DOI: 10.4049/jimmunol.1301576
Chronic follicular bronchiolitis requires antigen-specific regulatory T cell control to prevent fatal disease progression
Abstract
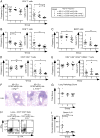
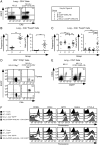
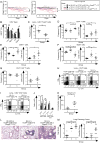
To study regulatory T (Treg) cell control of chronic autoimmunity in a lymphoreplete host, we created and characterized a new model of autoimmune lung inflammation that targets the medium and small airways. We generated transgenic mice that express a chimeric membrane protein consisting of hen egg lysozyme and a hemoglobin epitope tag under the control of the Clara cell secretory protein promoter, which largely limited transgene expression to the respiratory bronchioles. When Clara cell secretory protein-membrane hen egg lysozyme/hemoglobin transgenic mice were crossed to N3.L2 TCR transgenic mice that recognize the hemoglobin epitope, the bigenic progeny developed dense, pseudo-follicular lymphocytic peribronchiolar infiltrates that resembled the histological pattern of follicular bronchiolitis. Aggregates of activated IFN-γ- and IL-17A-secreting CD4(+) T cells as well as B cells surrounded the airways. Lung pathology was similar in Ifng(-/-) and Il17a(-/-) mice, indicating that either cytokine is sufficient to establish chronic disease. A large number of Ag-specific Treg cells accumulated in the lesions, and Treg cell depletion in the affected mice led to an interstitial spread of the disease that ultimately proved fatal. Thus, Treg cells act to restrain autoimmune responses, resulting in an organized and controlled chronic pathological process rather than a progressive disease.
Figures



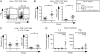

Similar articles
-
Regulatory T cells control epitope spreading in autoimmune arthritis independent of cytotoxic T-lymphocyte antigen-4.Immunology. 2018 Dec;155(4):446-457. doi: 10.1111/imm.12983. Epub 2018 Jul 31. Immunology. 2018. PMID: 29992549 Free PMC article.
-
Lung CD25 CD4 regulatory T cells suppress type 2 immune responses but not bronchial hyperreactivity.J Immunol. 2003 Jun 1;170(11):5502-10. doi: 10.4049/jimmunol.170.11.5502. J Immunol. 2003. PMID: 12759427
-
A threshold for central T cell tolerance to an inducible serum protein.J Immunol. 2003 Mar 15;170(6):3007-14. doi: 10.4049/jimmunol.170.6.3007. J Immunol. 2003. PMID: 12626554
-
Interleukin-23 is critical for full-blown expression of a non-autoimmune destructive arthritis and regulates interleukin-17A and RORgammat in gammadelta T cells.Arthritis Res Ther. 2009;11(6):R194. doi: 10.1186/ar2893. Epub 2009 Dec 17. Arthritis Res Ther. 2009. PMID: 20017902 Free PMC article.
-
B lymphocytes treated in vitro with antigen coupled to cholera toxin B subunit induce antigen-specific Foxp3(+) regulatory T cells and protect against experimental autoimmune encephalomyelitis.J Immunol. 2012 Feb 15;188(4):1686-97. doi: 10.4049/jimmunol.1101771. Epub 2012 Jan 16. J Immunol. 2012. PMID: 22250081
Cited by
-
The Clinical Characteristics and Outcomes of Follicular Bronchiolitis in Chinese Adult Patients.Sci Rep. 2018 May 8;8(1):7300. doi: 10.1038/s41598-018-25670-8. Sci Rep. 2018. PMID: 29740120 Free PMC article.
-
A model of TH17-associated ileal hyperplasia that requires both IL-17A and IFNγ to generate self-tolerance and prevent colitis.Mucosal Immunol. 2018 Jul;11(4):1127-1137. doi: 10.1038/s41385-018-0023-6. Epub 2018 May 4. Mucosal Immunol. 2018. PMID: 29728642 Free PMC article.
-
A human STAT3 gain-of-function variant confers T cell dysregulation without predominant Treg dysfunction in mice.JCI Insight. 2022 Nov 8;7(21):e162695. doi: 10.1172/jci.insight.162695. JCI Insight. 2022. PMID: 36136607 Free PMC article.
References
-
- Sakaguchi S, Sakaguchi N, Asano M, Itoh M, Toda M. Immunologic self-tolerance maintained by activated T cells expressing IL-2 receptor alpha-chains (CD25). Breakdown of a single mechanism of self-tolerance causes various autoimmune diseases. J Immunol. 1995;155:1151–1164. - PubMed
-
- Brunkow ME, Jeffery EW, Hjerrild KA, Paeper B, Clark LB, Yasayko SA, Wilkinson JE, Galas D, Ziegler SF, Ramsdell F. Disruption of a new forkhead/winged-helix protein, scurfin, results in the fatal lymphoproliferative disorder of the scurfy mouse. Nat Genet. 2001;27:68–73. - PubMed
-
- Bennett CL, Christie J, Ramsdell F, Brunkow ME, Ferguson PJ, Whitesell L, Kelly TE, Saulsbury FT, Chance PF, Ochs HD. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat Genet. 2001;27:20–21. - PubMed
-
- Wildin RS, Ramsdell F, Peake J, Faravelli F, Casanova JL, Buist N, Levy-Lahad E, Mazzella M, Goulet O, Perroni L, Bricarelli FD, Byrne G, McEuen M, Proll S, Appleby M, Brunkow ME. X-linked neonatal diabetes mellitus, enteropathy and endocrinopathy syndrome is the human equivalent of mouse scurfy. Nat Genet. 2001;27:18–20. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

