Soluble receptor for advanced glycation end products predicts 28-day mortality in critically ill patients with sepsis
- PMID: 24164543
- PMCID: PMC4044864
- DOI: 10.3109/00365513.2013.849357
Soluble receptor for advanced glycation end products predicts 28-day mortality in critically ill patients with sepsis
Abstract
Objective: Multiple biomarkers are used to assess sepsis severity and prognosis. Increased levels of the soluble receptor for advanced glycation end products (sRAGE) were previously observed in sepsis but also in end-organ injury without sepsis. We evaluated associations between sRAGE and (i) 28-day mortality, (ii) sepsis severity, and (iii) individual organ failure. Traditional biomarkers procalcitonin (PCT), C-reactive protein (CRP) and lactate served as controls.
Methods: sRAGE, PCT, CRP, and lactate levels were observed on days 1 (D1) and 3 (D3) in 54 septic patients. We also assessed the correlation between the biomarkers and acute respiratory distress syndrome (ARDS), acute kidney injury (AKI) and acute heart failure.
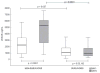
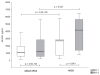
Results: There were 38 survivors and 16 non-survivors. On D1, non-survivors had higher sRAGE levels than survivors (p = 0.027). On D3, sRAGE further increased only in non-survivors (p < 0.0001) but remained unchanged in survivors. Unadjusted odds ratio (OR) for 28-day mortality was 8.2 (95% CI: 1.02-60.64) for sRAGE, p = 0.048. Receiver operating characteristic analysis determined strong correlation with outcome on D3 (AUC = 0.906, p < 0.001), superior to other studied biomarkers. sRAGE correlated with sepsis severity (p < 0.00001). sRAGE showed a significant positive correlation with PCT and CRP on D3. In patients without ARDS, sRAGE was significantly higher in non-survivors (p < 0.0001) on D3.
Conclusion: Increased sRAGE was associated with 28-day mortality in patients with sepsis, and was superior compared to PCT, CRP and lactate. sRAGE correlated with sepsis severity. sRAGE was increased in patients with individual organ failure. sRAGE could be used as an early biomarker in prognostication of outcome in septic patients.
Conflict of interest statement
Figures
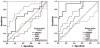


Similar articles
-
Plasma sRAGE is independently associated with increased mortality in ARDS: a meta-analysis of individual patient data.Intensive Care Med. 2018 Sep;44(9):1388-1399. doi: 10.1007/s00134-018-5327-1. Epub 2018 Jul 26. Intensive Care Med. 2018. PMID: 30051136 Free PMC article.
-
sRAGE as a Prognostic Biomarker in ARDS: Insights from a Clinical Cohort Study.Medicina (Kaunas). 2025 Jan 27;61(2):229. doi: 10.3390/medicina61020229. Medicina (Kaunas). 2025. PMID: 40005345 Free PMC article.
-
Soluble receptor for advanced glycation end products in critically ill patients and its associations with other clinical markers and 28-day mortality.Clin Interv Aging. 2014 Nov 19;9:1981-6. doi: 10.2147/CIA.S71130. eCollection 2014. Clin Interv Aging. 2014. PMID: 25429209 Free PMC article.
-
Plasma sRAGE Acts as a Genetically Regulated Causal Intermediate in Sepsis-associated Acute Respiratory Distress Syndrome.Am J Respir Crit Care Med. 2020 Jan 1;201(1):47-56. doi: 10.1164/rccm.201810-2033OC. Am J Respir Crit Care Med. 2020. PMID: 31487195 Free PMC article.
-
Is there any evidence that AGE/sRAGE is a universal biomarker/risk marker for diseases?Mol Cell Biochem. 2019 Jan;451(1-2):139-144. doi: 10.1007/s11010-018-3400-2. Epub 2018 Jun 30. Mol Cell Biochem. 2019. PMID: 29961210 Review.
Cited by
-
Plasma sRAGE is independently associated with increased mortality in ARDS: a meta-analysis of individual patient data.Intensive Care Med. 2018 Sep;44(9):1388-1399. doi: 10.1007/s00134-018-5327-1. Epub 2018 Jul 26. Intensive Care Med. 2018. PMID: 30051136 Free PMC article.
-
Pathophysiology of the Acute Respiratory Distress Syndrome: Insights from Clinical Studies.Crit Care Clin. 2021 Oct;37(4):795-815. doi: 10.1016/j.ccc.2021.05.005. Epub 2021 May 26. Crit Care Clin. 2021. PMID: 34548134 Free PMC article. Review.
-
The Course of Skin and Serum Biomarkers of Advanced Glycation Endproducts and Its Association with Oxidative Stress, Inflammation, Disease Severity, and Mortality during ICU Admission in Critically Ill Patients: Results from a Prospective Pilot Study.PLoS One. 2016 Aug 16;11(8):e0160893. doi: 10.1371/journal.pone.0160893. eCollection 2016. PLoS One. 2016. PMID: 27529340 Free PMC article.
-
Novel Perspectives Regarding the Pathology, Inflammation, and Biomarkers of Acute Respiratory Distress Syndrome.Int J Mol Sci. 2020 Dec 28;22(1):205. doi: 10.3390/ijms22010205. Int J Mol Sci. 2020. PMID: 33379178 Free PMC article. Review.
-
Prediction of mortality in adult patients with sepsis using six biomarkers: a systematic review and meta-analysis.Ann Intensive Care. 2019 Nov 8;9(1):125. doi: 10.1186/s13613-019-0600-1. Ann Intensive Care. 2019. PMID: 31705327 Free PMC article.
References
-
- Vincent JL, Donadello K, Schmit X. Biomarkers in the critically ill patient: C-reactive protein. Crit Care Clin. 2011;27:241–51. - PubMed
-
- Reinhart K, Meisner M. Biomarkers in the critically ill patient: procalcitonin. Crit Care Clin. 2011;27:253–63. - PubMed
-
- Hochreiter M, Schroeder S. Procalcitonin-based algorithm. Management of antibiotic therapy in critically ill patients. Anaesthesist. 2011;60:661–73. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
