Endocannabinoid anandamide mediates hypoxic pulmonary vasoconstriction
- PMID: 24167249
- PMCID: PMC3831960
- DOI: 10.1073/pnas.1308130110
Endocannabinoid anandamide mediates hypoxic pulmonary vasoconstriction
Abstract
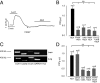
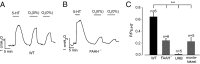
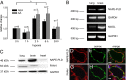
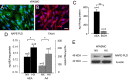
Endocannabinoids are important regulators of organ homeostasis. Although their role in systemic vasculature has been extensively studied, their impact on pulmonary vessels remains less clear. Herein, we show that the endocannabinoid anandamide (AEA) is a key mediator of hypoxic pulmonary vasoconstriction (HPV) via fatty acid amide hydrolase (FAAH)-dependent metabolites. This is underscored by the prominent vasoconstrictive effect of AEA on pulmonary arteries and strongly reduced HPV in FAAH(-/-) mice and wild-type mice upon pharmacological treatment with FAAH inhibitor URB597. In addition, mass spectrometry measurements revealed a clear increase of AEA and the FAAH-dependent metabolite arachidonic acid in hypoxic lungs of wild-type mice. We have identified pulmonary vascular smooth muscle cells as the source responsible for hypoxia-induced AEA generation. Moreover, either FAAH(-/-) mice or wild-type mice treated with FAAH inhibitor URB597 are protected against hypoxia-induced pulmonary hypertension and the concomitant vascular remodeling in the lung. Thus, the AEA/FAAH pathway is an important mediator of HPV and is involved in the generation of pulmonary hypertension.
Keywords: cannabinoid; pulmonary vascular tone.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Inhibition of fatty acid amide hydrolase activates Nrf2 signalling and induces heme oxygenase 1 transcription in breast cancer cells.Br J Pharmacol. 2013 Oct;170(3):489-505. doi: 10.1111/bph.12111. Br J Pharmacol. 2013. PMID: 23347118 Free PMC article.
-
Alterations in endocannabinoid tone following chemotherapy-induced peripheral neuropathy: effects of endocannabinoid deactivation inhibitors targeting fatty-acid amide hydrolase and monoacylglycerol lipase in comparison to reference analgesics following cisplatin treatment.Pharmacol Res. 2013 Jan;67(1):94-109. doi: 10.1016/j.phrs.2012.10.013. Epub 2012 Nov 2. Pharmacol Res. 2013. PMID: 23127915 Free PMC article.
-
Full inhibition of spinal FAAH leads to TRPV1-mediated analgesic effects in neuropathic rats and possible lipoxygenase-mediated remodeling of anandamide metabolism.PLoS One. 2013;8(4):e60040. doi: 10.1371/journal.pone.0060040. Epub 2013 Apr 3. PLoS One. 2013. PMID: 23573230 Free PMC article.
-
Amygdala FAAH and anandamide: mediating protection and recovery from stress.Trends Pharmacol Sci. 2013 Nov;34(11):637-44. doi: 10.1016/j.tips.2013.08.008. Epub 2013 Oct 25. Trends Pharmacol Sci. 2013. PMID: 24325918 Free PMC article. Review.
-
Anandamide and its metabolites: what are their roles in the kidney?Front Biosci (Schol Ed). 2016 Jun 1;8(2):264-77. doi: 10.2741/s461. Front Biosci (Schol Ed). 2016. PMID: 27100705 Free PMC article. Review.
Cited by
-
Cannabinoids in arterial, pulmonary and portal hypertension - mechanisms of action and potential therapeutic significance.Br J Pharmacol. 2019 May;176(10):1395-1411. doi: 10.1111/bph.14168. Epub 2018 Apr 14. Br J Pharmacol. 2019. PMID: 29455452 Free PMC article. Review.
-
A Systematic Review of the Complex Effects of Cannabinoids on Cerebral and Peripheral Circulation in Animal Models.Front Physiol. 2018 May 29;9:622. doi: 10.3389/fphys.2018.00622. eCollection 2018. Front Physiol. 2018. PMID: 29896112 Free PMC article. Review.
-
Effects of maternal edible THC consumption on offspring lung growth and function in a rhesus macaque model.Am J Physiol Lung Cell Mol Physiol. 2025 Mar 1;328(3):L463-L477. doi: 10.1152/ajplung.00360.2024. Epub 2025 Feb 4. Am J Physiol Lung Cell Mol Physiol. 2025. PMID: 39903192 Free PMC article.
-
Inhibition of Vascular Growth by Modulation of the Anandamide/Fatty Acid Amide Hydrolase Axis.Arterioscler Thromb Vasc Biol. 2021 Dec;41(12):2974-2989. doi: 10.1161/ATVBAHA.121.316973. Epub 2021 Oct 7. Arterioscler Thromb Vasc Biol. 2021. PMID: 34615374 Free PMC article.
-
Lack of hippocampal CB1 receptor desensitization by Δ(9)-tetrahydrocannabinol in aged mice and by low doses of JZL 184.Naunyn Schmiedebergs Arch Pharmacol. 2016 Jun;389(6):603-12. doi: 10.1007/s00210-016-1226-6. Epub 2016 Mar 17. Naunyn Schmiedebergs Arch Pharmacol. 2016. PMID: 26984820
References
-
- Gauthier KM, et al. Endothelium-derived 2-arachidonylglycerol: An intermediate in vasodilatory eicosanoid release in bovine coronary arteries. Am J Physiol Heart Circ Physiol. 2005;288(3):H1344–H1351. - PubMed
-
- Neumann P, Hedenstierna G. Ventilation-perfusion distributions in different porcine lung injury models. Acta Anaesthesiol Scand. 2001;45(1):78–86. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

