Structure and function of the Smoothened extracellular domain in vertebrate Hedgehog signaling
- PMID: 24171105
- PMCID: PMC3809587
- DOI: 10.7554/eLife.01340
Structure and function of the Smoothened extracellular domain in vertebrate Hedgehog signaling
Abstract
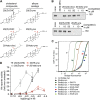
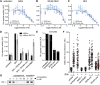
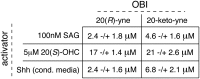
The Hedgehog (Hh) signal is transduced across the membrane by the heptahelical protein Smoothened (Smo), a developmental regulator, oncoprotein and drug target in oncology. We present the 2.3 Å crystal structure of the extracellular cysteine rich domain (CRD) of vertebrate Smo and show that it binds to oxysterols, endogenous lipids that activate Hh signaling. The oxysterol-binding groove in the Smo CRD is analogous to that used by Frizzled 8 to bind to the palmitoleyl group of Wnt ligands and to similar pockets used by other Frizzled-like CRDs to bind hydrophobic ligands. The CRD is required for signaling in response to native Hh ligands, showing that it is an important regulatory module for Smo activation. Indeed, targeting of the Smo CRD by oxysterol-inspired small molecules can block signaling by all known classes of Hh activators and by clinically relevant Smo mutants. DOI:http://dx.doi.org/10.7554/eLife.01340.001.
Keywords: Hedgehog signaling; Zebrafish; cysteine rich domain; oxysterol; smoothened.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures


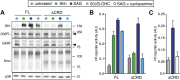
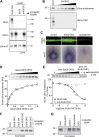
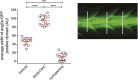
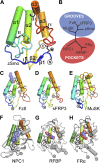
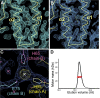

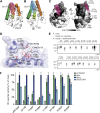
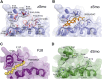

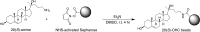



Comment in
-
A novel site comes into sight.Elife. 2013 Dec 3;2:e01680. doi: 10.7554/eLife.01680. Elife. 2013. PMID: 24302574 Free PMC article.
References
-
- Barbier P. 1899. Synthese du diethylheptenol. C R Hebd Seances Acad Sc 128:110
Publication types
MeSH terms
Substances
Grants and funding
- WT/082301/Z/07/Z/WT_/Wellcome Trust/United Kingdom
- R00 CA129174/CA/NCI NIH HHS/United States
- T32 HL007275/HL/NHLBI NIH HHS/United States
- 14414/CRUK_/Cancer Research UK/United Kingdom
- 090532/WT_/Wellcome Trust/United Kingdom
- C20724/A14414/CRUK_/Cancer Research UK/United Kingdom
- P30 DK020579/DK/NIDDK NIH HHS/United States
- P41 RR000954/RR/NCRR NIH HHS/United States
- P01 GM047969/GM/NIGMS NIH HHS/United States
- P30 DK056341/DK/NIDDK NIH HHS/United States
- P60 DK020579/DK/NIDDK NIH HHS/United States
- R01 HL067773/HL/NHLBI NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

