Direct observation of microtubule pushing by cortical dynein in living cells
- PMID: 24173713
- PMCID: PMC3873897
- DOI: 10.1091/mbc.E13-07-0376
Direct observation of microtubule pushing by cortical dynein in living cells
Abstract
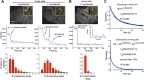
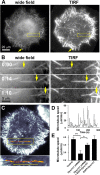
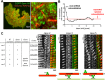
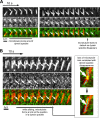
Microtubules are under the influence of forces mediated by cytoplasmic dynein motors associated with the cell cortex. If such microtubules are free to move, they are rapidly transported inside cells. Here we directly observe fluorescent protein-labeled cortical dynein speckles and motile microtubules. We find that several dynein complex subunits, including the heavy chain, the intermediate chain, and the associated dynactin subunit Dctn1 (also known as p150glued) form spatially resolved, dynamic speckles at the cell cortex, which are preferentially associated with microtubules. Measurements of bleaching and dissociation kinetics at the cell cortex reveal that these speckles often contain multiple labeled dynein heavy-chain molecules and turn over rapidly within seconds. The dynamic behavior of microtubules, such as directional movement, bending, or rotation, is influenced by association with dynein speckles, suggesting a direct physical and functional interaction. Our results support a model in which rapid turnover of cell cortex-associated dynein complexes facilitates their search to efficiently capture and push microtubules directionally with leading plus ends.
Figures
Comment in
-
Cytoskeletal self-organization in neuromorphogenesis.Bioarchitecture. 2014 Mar-Apr;4(2):75-80. doi: 10.4161/bioa.29070. Epub 2014 May 21. Bioarchitecture. 2014. PMID: 24847718 Free PMC article.
Similar articles
-
Analysis of the dynein-dynactin interaction in vitro and in vivo.Mol Biol Cell. 2003 Dec;14(12):5089-97. doi: 10.1091/mbc.e03-01-0025. Epub 2003 Oct 17. Mol Biol Cell. 2003. PMID: 14565986 Free PMC article.
-
Regulation of dynactin through the differential expression of p150Glued isoforms.J Biol Chem. 2008 Nov 28;283(48):33611-9. doi: 10.1074/jbc.M804840200. Epub 2008 Sep 22. J Biol Chem. 2008. PMID: 18812314 Free PMC article.
-
Dynactin functions as both a dynamic tether and brake during dynein-driven motility.Nat Commun. 2014 Sep 4;5:4807. doi: 10.1038/ncomms5807. Nat Commun. 2014. PMID: 25185702 Free PMC article.
-
[Dynein and dynactin as organizers of the system of cell microtubules].Ontogenez. 2006 Sep-Oct;37(5):323-39. Ontogenez. 2006. PMID: 17066975 Review. Russian.
-
"Search-and-capture" of microtubules through plus-end-binding proteins (+TIPs).J Biochem. 2003 Sep;134(3):321-6. doi: 10.1093/jb/mvg148. J Biochem. 2003. PMID: 14561716 Review.
Cited by
-
Establishing neuronal polarity: microtubule regulation during neurite initiation.Oxf Open Neurosci. 2022 May 13;1:kvac007. doi: 10.1093/oons/kvac007. eCollection 2022. Oxf Open Neurosci. 2022. PMID: 38596701 Free PMC article. Review.
-
Polarity sorting of axonal microtubules: a computational study.Mol Biol Cell. 2017 Nov 7;28(23):3271-3285. doi: 10.1091/mbc.E17-06-0380. Epub 2017 Oct 4. Mol Biol Cell. 2017. PMID: 28978741 Free PMC article.
-
Moonlighting Motors: Kinesin, Dynein, and Cell Polarity.Trends Cell Biol. 2017 Jul;27(7):505-514. doi: 10.1016/j.tcb.2017.02.005. Epub 2017 Mar 8. Trends Cell Biol. 2017. PMID: 28284467 Free PMC article. Review.
-
Crosstalk of cell polarity signaling pathways.Protoplasma. 2017 May;254(3):1241-1258. doi: 10.1007/s00709-017-1075-2. Epub 2017 Mar 14. Protoplasma. 2017. PMID: 28293820 Review.
-
Cell adhesion and spreading on fluid membranes through microtubules-dependent mechanotransduction.Nat Commun. 2025 Jan 31;16(1):1201. doi: 10.1038/s41467-025-56343-6. Nat Commun. 2025. PMID: 39885125 Free PMC article.
References
-
- Ahmad FJ, He Y, Myers KA, Hasaka TP, Francis F, Black MM, Baas PW. Effects of dynactin disruption and dynein depletion on axonal microtubules. Traffic. 2006;7:524–537. - PubMed
-
- Ahmad FJ, Hughey J, Wittmann T, Hyman A, Greaser M, Baas PW. Motor proteins regulate force interactions between microtubules and microfilaments in the axon. Nat Cell Biol. 2000;2:276–280. - PubMed
-
- Dehmelt L, Bastiaens PI. Spatial organization of intracellular communication: insights from imaging. Nat Rev Mol Cell Biol. 2010;11:440–452. - PubMed
-
- Dehmelt L, Bastiaens P. Self-organization in cells. In: Meyer-Ortmanns H, Thurner S, editors. In: Principles of Evolution. Heidelberg, Germany: Springer; 2011. pp. 219–238.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

