Pericytes synthesize renin
- PMID: 24175260
- PMCID: PMC3782206
- DOI: 10.5527/wjn.v2.i1.11
Pericytes synthesize renin
Abstract
Aim: To investigate renin expression in pericytes during normal kidney development and after deletion of angiotensinogen, the precursor for all angiotensins.
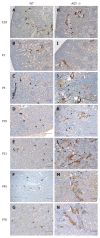
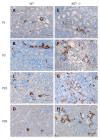
Methods: We examined the distribution of renin expressing cells by immunoshistochemistry in the interstitial compartment of wild type (WT) and angiotensinogen deficient (AGT -/-) mice at different developmental stages from embryonic day 18 (E18: WT, n = 4; AGT -/-, n = 5) and at day 1 (P1: WT, n = 5; AGT -/-, n = 5), 5 (P5: WT, n = 7; AGT -/-, n = 8), 10 (P10: WT, n = 3; AGT -/-, n = 5), 21 (P21: WT, n = 7; AGT -/-, n = 5), 45 (P45: WT, n = 3; AGT -/-, n = 3), and 70 (P70: WT, n = 2; AGT -/-, n = 2) of postnatal life. We quantified the number of pericytes positive for renin at all the developmental stages mentioned above and compared the results of AGT -/- mice to their WT counterparts.
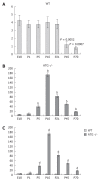
Results: In WT mice, renal interstitial pericytes synthesize renin in early life supporting a lineage relationship with renin cells in the vasculature. The number of pericytes positive for renin per area of 0.32 mm(2) (density) in WT mice was maintained from fetal life till weaning age (E18 = 4.25 ± 0.63, P1 = 3.75 ± 0.48, P5 = 3.75 ± 0.48, P10 = 4 ± 0.71, P21 = 3.8 ± 0.58) and markedly decreased in adult life (P45 = 1.2 ± 0.37, P70 = 0.8 ± 0.20). On the other hand, in AGT -/- mice the density of pericytes expressing renin was not significantly different from WT mice at E18 and P1: E18 = 5.75 ± 0.50 vs 4.25 ± 0.63 (P = 0.106), P1 = 9.25 ± 3.50 vs 3.75 ± 0.48 (P = 0.175) but significantly increased from P5 till P70: P5 = 38.25 ± 5 vs 3.75 ± 0.48 (P = 0.0004), P10 = 173 ± 7.50 vs 4 ± 0.70 (P = 5.24567 × 10(-7)), P21 = 83 ± 6.70 vs 3.8 ± 0.58 (P = 2.97358 × 10(-6)), P45 = 49 ± 3.50 vs 1.2 ± 0.37 (P = 8.18274 x 10(-7)) and P70 = 17.8 ± 2.30 vs 0.8 ± 0.20 (P = 3.51151 × 10(-5)). The AGT -/- mice showed a marked increase in the number of pericytes per field studied starting from P5, reaching its peak at P10, and then a gradually decreasing until P70.
Conclusion: Interstitial pericytes synthesize renin during development and the number of renin-expressing pericytes increases in response to a homeostatic threat imposed early in life such as lack of angiotensinogen.
Keywords: Angiotensin deficiency; Angiotensinogen; Development; Gene deletion; Homeostasis; Interstitium; Kidney; Renin angiotensin system.
Figures
References
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

