Pharmacokinetics of mirabegron, a β3-adrenoceptor agonist for treatment of overactive bladder, in healthy Japanese male subjects: results from single- and multiple-dose studies
- PMID: 24178236
- PMCID: PMC3880470
- DOI: 10.1007/s40261-013-0146-1
Pharmacokinetics of mirabegron, a β3-adrenoceptor agonist for treatment of overactive bladder, in healthy Japanese male subjects: results from single- and multiple-dose studies
Abstract
Background: Mirabegron is a human β3-adrenoceptor agonist for the treatment of overactive bladder. The pharmacokinetic profile of mirabegron has been extensively characterized in healthy Caucasian subjects.
Objective: The objective of this study was to evaluate the pharmacokinetics, dose-proportionality, and tolerability of mirabegron following single and multiple oral doses in healthy Japanese male subjects. The results were compared with those reported in non-Japanese (primarily Caucasian) subjects.
Methods: Two studies were conducted. In a single-blind, randomized, placebo-controlled, parallel-group, single- and multiple-ascending dose study (Study 1), mirabegron oral controlled absorption system (OCAS) tablets were administered at single doses of 50, 100, 200, 300, and 400 mg, with eight subjects (six active, two placebo) per dose group (Part I), and once daily for 7 days at 100 and 200 mg with 12 subjects (eight active, four placebo) per group (Part II). In an open-label, three-period, single-ascending dose study (Study 2), mirabegron OCAS was administered to 12 subjects at 25, 50, and 100 mg in an intra-subject dose-escalation design. Plasma and/or urine samples were collected up to 72 h after the first and last dose and analyzed for mirabegron. Pharmacokinetic parameters were determined using non-compartmental methods. Tolerability assessments included physical examinations, vital signs, 12-lead electrocardiogram, clinical laboratory tests (biochemistry, hematology, and urinalysis), and adverse event (AE) monitoring.
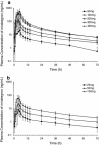
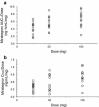
Results: Forty and 24 young male subjects completed Part I and II, respectively, of Study 1. Twelve young males completed Study 2. After single oral doses (25-400 mg), maximum plasma concentrations (C max) were reached at approximately 2.8-4.0 h postdose. Plasma exposure (C max and area under the plasma concentration-time curve) of mirabegron increased more than dose proportionally at single doses of 25-100 mg and approximately dose proportionally at high doses of 300 and 400 mg. A more than dose proportional increase in plasma exposure was noted in the body of the same individual. Mirabegron accumulated twofold upon once-daily dosing relative to single-dose data. Steady state was reached within 7 days. Mirabegron was generally well-tolerated at single doses up to 400 mg and multiple doses up to 200 mg. The AE with the highest incidence was increased pulse rate at 400 mg in Study 1.
Conclusions: Mirabegron OCAS exhibits similar single- and multiple-dose pharmacokinetic characteristics and deviations from dose proportionality in healthy Japanese male subjects compared with those observed in non-Japanese (primarily Caucasian) subjects in previous studies.
Figures


References
-
- Astellas Pharma. Betanis tablets Japanese prescribing information. July 2011.http://www.info.pmda.go.jp/shinyaku/P201100119/80012600_22300AMX00592_A1.... Accessed 29 Oct 2013
-
- Astellas Pharma US Inc. Myrbetriq United States prescribing information. June 2012. http://www.accessdata.fda.gov/scripts/cder/drugsatfda/index.cfm?fuseacti.... Accessed 29 Oct 2013
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

