Resveratrol induces a mitochondrial complex I-dependent increase in NADH oxidation responsible for sirtuin activation in liver cells
- PMID: 24178296
- PMCID: PMC3868777
- DOI: 10.1074/jbc.M113.466490
Resveratrol induces a mitochondrial complex I-dependent increase in NADH oxidation responsible for sirtuin activation in liver cells
Abstract
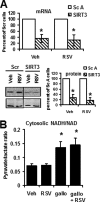
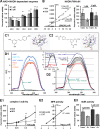
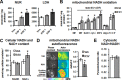
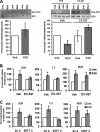
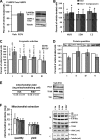
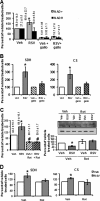
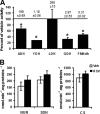
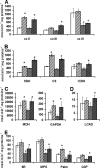
Resveratrol (RSV) has been shown to be involved in the regulation of energetic metabolism, generating increasing interest in therapeutic use. SIRT1 has been described as the main target of RSV. However, recent reports have challenged the hypothesis of its direct activation by RSV, and the signaling pathways remain elusive. Here, the effects of RSV on mitochondrial metabolism are detailed both in vivo and in vitro using murine and cellular models and isolated enzymes. We demonstrate that low RSV doses (1-5 μM) directly stimulate NADH dehydrogenases and, more specifically, mitochondrial complex I activity (EC50 ∼1 μM). In HepG2 cells, this complex I activation increases the mitochondrial NAD(+)/NADH ratio. This higher NAD(+) level initiates a SIRT3-dependent increase in the mitochondrial substrate supply pathways (i.e. the tricarboxylic acid cycle and fatty acid oxidation). This effect is also seen in liver mitochondria of RSV-fed animals (50 mg/kg/day). We conclude that the increase in NADH oxidation by complex I is a crucial event for SIRT3 activation by RSV. Our results open up new perspectives in the understanding of the RSV signaling pathway and highlight the critical importance of RSV doses used for future clinical trials.
Keywords: Complex I; Mitochondria; Mitochondrial Metabolism; NAD; NADH Dehydrogenase; Resveratrol; Sirtuins.
Figures

Similar articles
-
Mechanisms of cell death pathway activation following drug-induced inhibition of mitochondrial complex I.Redox Biol. 2015;4:279-88. doi: 10.1016/j.redox.2015.01.005. Epub 2015 Jan 16. Redox Biol. 2015. PMID: 25625582 Free PMC article.
-
Sirtuin 3 is required for the protective effect of Resveratrol on Manganese-induced disruption of mitochondrial biogenesis in primary cultured neurons.J Neurochem. 2021 Jan;156(1):121-135. doi: 10.1111/jnc.15095. Epub 2020 Jun 16. J Neurochem. 2021. PMID: 32426865
-
Resveratrol attenuates oxidative stress in mitochondrial Complex I deficiency: Involvement of SIRT3.Free Radic Biol Med. 2016 Jul;96:190-8. doi: 10.1016/j.freeradbiomed.2016.04.027. Epub 2016 Apr 25. Free Radic Biol Med. 2016. PMID: 27126960
-
Interplay between NADH oxidation by complex I, glutathione redox state and sirtuin-3, and its role in the development of insulin resistance.Biochim Biophys Acta Mol Basis Dis. 2020 Aug 1;1866(8):165801. doi: 10.1016/j.bbadis.2020.165801. Epub 2020 Apr 16. Biochim Biophys Acta Mol Basis Dis. 2020. PMID: 32305451 Review.
-
Dosis Facit Sanitatem-Concentration-Dependent Effects of Resveratrol on Mitochondria.Nutrients. 2017 Oct 13;9(10):1117. doi: 10.3390/nu9101117. Nutrients. 2017. PMID: 29027961 Free PMC article. Review.
Cited by
-
Targeting Mitochondrial Dysfunction for the Prevention and Treatment of Metabolic Disease by Bioactive Food Components.J Lipid Atheroscler. 2024 Sep;13(3):306-327. doi: 10.12997/jla.2024.13.3.306. Epub 2024 Jun 17. J Lipid Atheroscler. 2024. PMID: 39355406 Free PMC article. Review.
-
Regulation of Cytochrome c Oxidase by Natural Compounds Resveratrol, (-)-Epicatechin, and Betaine.Cells. 2021 May 29;10(6):1346. doi: 10.3390/cells10061346. Cells. 2021. PMID: 34072396 Free PMC article. Review.
-
Resveratrol Improved Flow-Mediated Outward Arterial Remodeling in Ovariectomized Rats with Hypertrophic Effect at High Dose.PLoS One. 2016 Jan 6;11(1):e0146148. doi: 10.1371/journal.pone.0146148. eCollection 2016. PLoS One. 2016. PMID: 26734763 Free PMC article.
-
Mitochondrial Dysfunction as a Pathogenesis and Therapeutic Strategy for Metabolic-Dysfunction-Associated Steatotic Liver Disease.Int J Mol Sci. 2025 Apr 30;26(9):4256. doi: 10.3390/ijms26094256. Int J Mol Sci. 2025. PMID: 40362504 Free PMC article. Review.
-
Characterization of the 5'-flanking region of the human DNA helicase B (HELB) gene and its response to trans-Resveratrol.Sci Rep. 2016 Apr 15;6:24510. doi: 10.1038/srep24510. Sci Rep. 2016. PMID: 27079536 Free PMC article.
References
-
- Richard T., Pawlus A. D., Iglesias M. L., Pedrot E., Waffo-Teguo P., Merillon J. M., Monti J. P. (2001) Neuroprotective properties of resveratrol and derivatives Ann. N.Y. Acad. Sci. 1215, 103–108 - PubMed
-
- Petrovski G., Gurusamy N., Das D. K. (2011) Resveratrol in cardiovascular health and disease Ann. N.Y. Acad. Sci. 1215, 22–33 - PubMed
-
- Baur J. A., Pearson K. J., Price N. L., Jamieson H. A., Lerin C., Kalra A., Prabhu V. V., Allard J. S., Lopez-Lluch G., Lewis K., Pistell P. J., Poosala S., Becker K. G., Boss O., Gwinn D., Wang M., Ramaswamy S., Fishbein K. W., Spencer R. G., Lakatta E. G., Le Couteur D., Shaw R. J., Navas P., Puigserver P., Ingram D. K., de Cabo R., Sinclair D. A. (2006) Resveratrol improves health and survival of mice on a high-calorie diet. Nature 444, 337–342 - PMC - PubMed
-
- Lagouge M., Argmann C., Gerhart-Hines Z., Meziane H., Lerin C., Daussin F., Messadeq N., Milne J., Lambert P., Elliott P., Geny B., Laakso M., Puigserver P., Auwerx J. (2006) Resveratrol improves mitochondrial function and protects against metabolic disease by activating SIRT1 and PGC-1α. Cell 127, 1109–1122 - PubMed
-
- Mercader J., Palou A., Bonet M. L. (2011) Resveratrol enhances fatty acid oxidation capacity and reduces resistin and retinol-binding protein 4 expression in white adipocytes. J. Nutr. Biochem. 22, 828–834 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

