New insights into mechanisms controlling the NLRP3 inflammasome and its role in lung disease
- PMID: 24183846
- PMCID: PMC3873477
- DOI: 10.1016/j.ajpath.2013.09.007
New insights into mechanisms controlling the NLRP3 inflammasome and its role in lung disease
Abstract
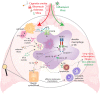
Inflammasomes are large macromolecular signaling complexes that control the proteolytic activation of two highly proinflammatory IL-1 family cytokines, IL-1β and IL-18. The NLRP3 inflammasome is of special interest because it can assemble in response to a diverse array of stimuli and because the inflammation it triggers has been implicated in a wide variety of disease pathologies. To avoid aberrant activation, the NLRP3 inflammasome is modulated on multiple levels, ranging from transcriptional control to post-translational protein modifications. Emerging genetic and pharmacological evidence suggests that NLRP3 inflammasome activation may also be involved in acute lung inflammation after viral infection and during progression of several chronic pulmonary diseases, including idiopathic pulmonary fibrosis, chronic obstructive pulmonary disease, and asthma. Here, we review the most recent contributions to our understanding of the regulatory mechanisms controlling activation of the NLRP3 inflammasome and discuss the contribution of the NLRP3 inflammasome to the pathology of lung diseases.
Copyright © 2014 American Society for Investigative Pathology. Published by Elsevier Inc. All rights reserved.
Figures

References
-
- Toews G.B. Cytokines and the lung. Eur Respir J Suppl. 2001;34:3s–17s. - PubMed
-
- Martinon F., Burns K., Tschopp J. The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-beta. Mol Cell. 2002;10:417–426. - PubMed
-
- Bousquet J., Khaltaev N., editors. Global surveillance, prevention and control of chronic respiratory diseases: a comprehensive approach. WHO Press; Geneva: 2007. pp v, 15, 21.
-
- Dinarello C.A. Immunological and inflammatory functions of the interleukin-1 family. Annu Rev Immunol. 2009;27:519–550. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

