Optimisation of engineered Escherichia coli biofilms for enzymatic biosynthesis of l-halotryptophans
- PMID: 24188712
- PMCID: PMC3843566
- DOI: 10.1186/2191-0855-3-66
Optimisation of engineered Escherichia coli biofilms for enzymatic biosynthesis of l-halotryptophans
Abstract
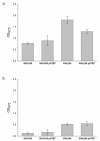
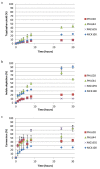
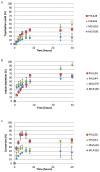
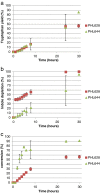
Engineered biofilms comprising a single recombinant species have demonstrated remarkable activity as novel biocatalysts for a range of applications. In this work, we focused on the biotransformation of 5-haloindole into 5-halotryptophan, a pharmaceutical intermediate, using Escherichia coli expressing a recombinant tryptophan synthase enzyme encoded by plasmid pSTB7. To optimise the reaction we compared two E. coli K-12 strains (MC4100 and MG1655) and their ompR234 mutants, which overproduce the adhesin curli (PHL644 and PHL628). The ompR234 mutation increased the quantity of biofilm in both MG1655 and MC4100 backgrounds. In all cases, no conversion of 5-haloindoles was observed using cells without the pSTB7 plasmid. Engineered biofilms of strains PHL628 pSTB7 and PHL644 pSTB7 generated more 5-halotryptophan than their corresponding planktonic cells. Flow cytometry revealed that the vast majority of cells were alive after 24 hour biotransformation reactions, both in planktonic and biofilm forms, suggesting that cell viability was not a major factor in the greater performance of biofilm reactions. Monitoring 5-haloindole depletion, 5-halotryptophan synthesis and the percentage conversion of the biotransformation reaction suggested that there were inherent differences between strains MG1655 and MC4100, and between planktonic and biofilm cells, in terms of tryptophan and indole metabolism and transport. The study has reinforced the need to thoroughly investigate bacterial physiology and make informed strain selections when developing biotransformation reactions.
Figures


Similar articles
-
Non-pathogenic Escherichia coli biofilms: effects of growth conditions and surface properties on structure and curli gene expression.Arch Microbiol. 2020 Aug;202(6):1517-1527. doi: 10.1007/s00203-020-01864-5. Epub 2020 Mar 28. Arch Microbiol. 2020. PMID: 32222779 Free PMC article.
-
Rapid enzyme regeneration results in the striking catalytic longevity of an engineered, single species, biocatalytic biofilm.Microb Cell Fact. 2016 Oct 21;15(1):180. doi: 10.1186/s12934-016-0579-3. Microb Cell Fact. 2016. PMID: 27769259 Free PMC article.
-
Polymer-induced biofilms for enhanced biocatalysis.Mater Horiz. 2022 Oct 3;9(10):2592-2602. doi: 10.1039/d2mh00607c. Mater Horiz. 2022. PMID: 35912866 Free PMC article.
-
The curli biosynthesis regulator CsgD co-ordinates the expression of both positive and negative determinants for biofilm formation in Escherichia coli.Microbiology (Reading). 2003 Oct;149(Pt 10):2847-2857. doi: 10.1099/mic.0.26306-0. Microbiology (Reading). 2003. PMID: 14523117
-
Escherichia coli serotype O157:H7 retention on solid surfaces and peroxide resistance is enhanced by dual-strain biofilm formation.Foodborne Pathog Dis. 2010 Aug;7(8):935-43. doi: 10.1089/fpd.2009.0503. Foodborne Pathog Dis. 2010. PMID: 20367070
Cited by
-
Biofilm architecture: An emerging synthetic biology target.Synth Syst Biotechnol. 2020 Jan 13;5(1):1-10. doi: 10.1016/j.synbio.2020.01.001. eCollection 2020 Mar. Synth Syst Biotechnol. 2020. PMID: 31956705 Free PMC article. Review.
-
Unlocking Reactivity of TrpB: A General Biocatalytic Platform for Synthesis of Tryptophan Analogues.J Am Chem Soc. 2017 Aug 9;139(31):10769-10776. doi: 10.1021/jacs.7b05007. Epub 2017 Jul 28. J Am Chem Soc. 2017. PMID: 28708383 Free PMC article.
-
A Panel of TrpB Biocatalysts Derived from Tryptophan Synthase through the Transfer of Mutations that Mimic Allosteric Activation.Angew Chem Int Ed Engl. 2016 Sep 12;55(38):11577-81. doi: 10.1002/anie.201606242. Epub 2016 Aug 11. Angew Chem Int Ed Engl. 2016. PMID: 27510733 Free PMC article.
-
Non-pathogenic Escherichia coli biofilms: effects of growth conditions and surface properties on structure and curli gene expression.Arch Microbiol. 2020 Aug;202(6):1517-1527. doi: 10.1007/s00203-020-01864-5. Epub 2020 Mar 28. Arch Microbiol. 2020. PMID: 32222779 Free PMC article.
-
Hyperadherence of Pseudomonas taiwanensis VLB120ΔC increases productivity of (S)-styrene oxide formation.Microb Biotechnol. 2017 Jul;10(4):735-744. doi: 10.1111/1751-7915.12378. Epub 2016 Jul 14. Microb Biotechnol. 2017. PMID: 27411543 Free PMC article.
References
-
- Bhowmick PP, Devegowda D, Ruwandeepika HAD, Fuchs TM, Srikumar S, Karunasagar I, Karunasagar I. gcpA (stm1987) is critical for cellulose production and biofilm formation on polystyrene surface by Salmonella enterica serovar Weltevreden in both high and low nutrient medium. Microb Pathog. 2011;3:114–122. doi: 10.1016/j.micpath.2010.12.002. - DOI - PubMed
-
- Chu WH, Zere TR, Weber MM, Wood TK, Whiteley M, Hidalgo-Romano B, Valenzuela E, Mclean RJC. Indole Production Promotes Escherichia coli Mixed-Culture Growth with Pseudomonas aeruginosa by Inhibiting Quorum Signaling. Appl Environ Microbiol. 2012;3:411–419. doi: 10.1128/AEM.06396-11. - DOI - PMC - PubMed
-
- Cortes-Lorenzo C, Rodriguez-Diaz M, Lopez-Lopez C, Sanchez-Peinado M, Rodelas B, Gonzalez-Lopez J. Effect of salinity on enzymatic activities in a submerged fixed bed biofilm reactor for municipal sewage treatment. Bioresour Technol. 2012;3:312–319. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

