Intracellular ATP binding is required to activate the slowly activating K+ channel I(Ks)
- PMID: 24190995
- PMCID: PMC3839694
- DOI: 10.1073/pnas.1315649110
Intracellular ATP binding is required to activate the slowly activating K+ channel I(Ks)
Abstract
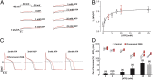
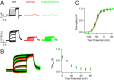
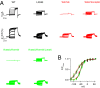
Gating of ion channels by ligands is fundamental to cellular function, and ATP serves as both an energy source and a signaling molecule that modulates ion channel and transporter functions. The slowly activating K(+) channel I(Ks) in cardiac myocytes is formed by KCNQ1 and KCNE1 subunits that conduct K(+) to repolarize the action potential. Here we show that intracellular ATP activates heterologously coexpressed KCNQ1 and KCNE1 as well as I(Ks) in cardiac myocytes by directly binding to the C terminus of KCNQ1 to allow the pore to open. The channel is most sensitive to ATP near its physiological concentration, and lowering ATP concentration in cardiac myocytes results in I(Ks) reduction and action potential prolongation. Multiple mutations that suppress I(Ks) by decreasing the ATP sensitivity of the channel are associated with the long QT (interval between the Q and T waves in electrocardiogram) syndrome that predisposes afflicted individuals to cardiac arrhythmia and sudden death. A cluster of basic and aromatic residues that may form a unique ATP binding site are identified; ATP activation of the wild-type channel and the effects of the mutations on ATP sensitivity are consistent with an allosteric mechanism. These results demonstrate the activation of an ion channel by intracellular ATP binding, and ATP-dependent gating allows I(Ks) to couple myocyte energy state to its electrophysiology in physiologic and pathologic conditions.
Keywords: heart failure; ischemia.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
The I Ks Ion Channel Activator Mefenamic Acid Requires KCNE1 and Modulates Channel Gating in a Subunit-Dependent Manner.Mol Pharmacol. 2020 Feb;97(2):132-144. doi: 10.1124/mol.119.117952. Epub 2019 Nov 13. Mol Pharmacol. 2020. PMID: 31722973
-
Ginsenoside Rg3 activates human KCNQ1 K+ channel currents through interacting with the K318 and V319 residues: a role of KCNE1 subunit.Eur J Pharmacol. 2010 Jul 10;637(1-3):138-47. doi: 10.1016/j.ejphar.2010.04.001. Epub 2010 Apr 21. Eur J Pharmacol. 2010. PMID: 20399767
-
Structural basis of slow activation gating in the cardiac I Ks channel complex.Cell Physiol Biochem. 2011;27(5):443-52. doi: 10.1159/000329965. Epub 2011 Jun 15. Cell Physiol Biochem. 2011. PMID: 21691061
-
KCNE1 and KCNE3: The yin and yang of voltage-gated K(+) channel regulation.Gene. 2016 Jan 15;576(1 Pt 1):1-13. doi: 10.1016/j.gene.2015.09.059. Epub 2015 Sep 26. Gene. 2016. PMID: 26410412 Free PMC article. Review.
-
Structural basis for K(V)7.1-KCNE(x) interactions in the I(Ks) channel complex.Heart Rhythm. 2010 May;7(5):708-13. doi: 10.1016/j.hrthm.2009.12.017. Epub 2009 Dec 24. Heart Rhythm. 2010. PMID: 20206317 Free PMC article. Review.
Cited by
-
Two-stage electro-mechanical coupling of a KV channel in voltage-dependent activation.Nat Commun. 2020 Feb 3;11(1):676. doi: 10.1038/s41467-020-14406-w. Nat Commun. 2020. PMID: 32015334 Free PMC article.
-
Arrhythmogenic cardiomyopathy in a patient with a rare loss-of-function KCNQ1 mutation.J Am Heart Assoc. 2015 Jan 23;4(1):e001526. doi: 10.1161/JAHA.114.001526. J Am Heart Assoc. 2015. PMID: 25616976 Free PMC article.
-
A long QT mutation substitutes cholesterol for phosphatidylinositol-4,5-bisphosphate in KCNQ1 channel regulation.PLoS One. 2014 Mar 28;9(3):e93255. doi: 10.1371/journal.pone.0093255. eCollection 2014. PLoS One. 2014. PMID: 24681627 Free PMC article.
-
Adverse remodeling of the electrophysiological response to ischemia-reperfusion in human heart failure is associated with remodeling of metabolic gene expression.Circ Arrhythm Electrophysiol. 2014 Oct;7(5):875-82. doi: 10.1161/CIRCEP.113.001477. Epub 2014 Aug 11. Circ Arrhythm Electrophysiol. 2014. PMID: 25114062 Free PMC article.
-
The KCNQ1 channel - remarkable flexibility in gating allows for functional versatility.J Physiol. 2015 Jun 15;593(12):2605-15. doi: 10.1113/jphysiol.2014.287607. Epub 2015 Mar 18. J Physiol. 2015. PMID: 25653179 Free PMC article. Review.
References
-
- Krebs EG. Historical perspectives on protein phosphorylation and a classification system for protein kinases. Philos Trans R Soc Lond B Biol Sci. 1983;302(1108):3–11. - PubMed
-
- Hilgemann DW. Cytoplasmic ATP-dependent regulation of ion transporters and channels: Mechanisms and messengers. Annu Rev Physiol. 1997;59:193–220. - PubMed
-
- Nichols CG, Lopatin AN. Inward rectifier potassium channels. Annu Rev Physiol. 1997;59:171–191. - PubMed
-
- Ashcroft SJ, Ashcroft FM. Properties and functions of ATP-sensitive K-channels. Cell Signal. 1990;2(3):197–214. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

