Improved tumor oxygenation and survival in glioblastoma patients who show increased blood perfusion after cediranib and chemoradiation
- PMID: 24190997
- PMCID: PMC3839699
- DOI: 10.1073/pnas.1318022110
Improved tumor oxygenation and survival in glioblastoma patients who show increased blood perfusion after cediranib and chemoradiation
Abstract
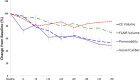
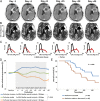
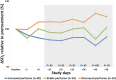
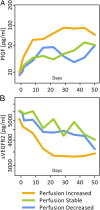
Antiangiogenic therapy has shown clear activity and improved survival benefit for certain tumor types. However, an incomplete understanding of the mechanisms of action of antiangiogenic agents has hindered optimization and broader application of this new therapeutic modality. In particular, the impact of antiangiogenic therapy on tumor blood flow and oxygenation status (i.e., the role of vessel pruning versus normalization) remains controversial. This controversy has become critical as multiple phase III trials of anti-VEGF agents combined with cytotoxics failed to show overall survival benefit in newly diagnosed glioblastoma (nGBM) patients and several other cancers. Here, we shed light on mechanisms of nGBM response to cediranib, a pan-VEGF receptor tyrosine kinase inhibitor, using MRI techniques and blood biomarkers in prospective phase II clinical trials of cediranib with chemoradiation vs. chemoradiation alone in nGBM patients. We demonstrate that improved perfusion occurs only in a subset of patients in cediranib-containing regimens, and is associated with improved overall survival in these nGBM patients. Moreover, an increase in perfusion is associated with improved tumor oxygenation status as well as with pharmacodynamic biomarkers, such as changes in plasma placenta growth factor and sVEGFR2. Finally, treatment resistance was associated with elevated plasma IL-8 and sVEGFR1 posttherapy. In conclusion, tumor perfusion changes after antiangiogenic therapy may distinguish responders vs. nonresponders early in the course of this expensive and potentially toxic form of therapy, and these results may provide new insight into the selection of glioblastoma patients most likely to benefit from anti-VEGF treatments.
Keywords: brain tumor; personalized treatment.
Conflict of interest statement
Conflict of interest statement: T.T.B. received consulting honoraria from Merck & Co., Roche, Kirin Pharmaceuticals, Spectrum, Amgen, and Novartis; has provided CME lectures and material for Up to Date, Inc., Robert Michael Educational Institute LLC, Educational Concepts Group, Research to Practice, Oakstone Medical Publishing, American Society of Hematology, and Imedex; has consulted for Champions Biotechnology, Advance Medical; and received research support from Pfizer, Astra Zeneca, and Millennium. E.R.G. received research support from Merck. D.G.D. served as a consultant for Hexal/Sandoz. R.K.J. received research grants from MedImmune and Roche; received consultant fees from Enlight, Noxxon, and Zyngenia; owns equity in Enlight, SynDevRx, and XTuit; and serves on the Board of Directors of XTuit and Boards of Trustees of H&Q Healthcare Investors and H&Q Life Sciences Investors.
Figures
Comment in
-
Targeted therapies: early vessel normalization improves glioblastoma outcomes.Nat Rev Clin Oncol. 2014 Jan;11(1):4. doi: 10.1038/nrclinonc.2013.228. Epub 2013 Nov 26. Nat Rev Clin Oncol. 2014. PMID: 24281060 No abstract available.
References
-
- CBTRUS (2012) CBTRUS Statistical Report: Primary Brain and Central Nervous System Tumors Diagnosed in the United States in 2004-2008 (March 23, 2010 Revision) (Central Brain Tumor Registry of the United States, Hinsdale, IL). Available at www.cbtrus.org. Accessed October 1, 2013.
-
- Stupp R, et al. European Organisation for Research and Treatment of Cancer Brain Tumor and Radiotherapy Groups National Cancer Institute of Canada Clinical Trials Group Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352(10):987–996. - PubMed
-
- Jain RK. Normalization of tumor vasculature: An emerging concept in antiangiogenic therapy. Science. 2005;307(5706):58–62. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- 5R01NS060918/NS/NINDS NIH HHS/United States
- R01CA159258/CA/NCI NIH HHS/United States
- 2013069/PHS HHS/United States
- R01 CA163815/CA/NCI NIH HHS/United States
- 1U01CA154601/CA/NCI NIH HHS/United States
- P01 CA080124/CA/NCI NIH HHS/United States
- R01CA129371/CA/NCI NIH HHS/United States
- U01 CA154601/CA/NCI NIH HHS/United States
- R01 CA159258/CA/NCI NIH HHS/United States
- R01CA163815/CA/NCI NIH HHS/United States
- R01 NS060918/NS/NINDS NIH HHS/United States
- P01CA080124/CA/NCI NIH HHS/United States
- P50 CA165962/CA/NCI NIH HHS/United States
- 8UL1TR000170-05/TR/NCATS NIH HHS/United States
- N01CM-2008-00060C/CM/NCI NIH HHS/United States
- R01 CA129371/CA/NCI NIH HHS/United States
- K24 CA125440/CA/NCI NIH HHS/United States
- K24CA125440A/CA/NCI NIH HHS/United States
- UL1 TR000170/TR/NCATS NIH HHS/United States
- 191088/V50/PHS HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

