Focal targeting by human β-defensin 2 disrupts localized virulence factor assembly sites in Enterococcus faecalis
- PMID: 24191013
- PMCID: PMC3864318
- DOI: 10.1073/pnas.1319066110
Focal targeting by human β-defensin 2 disrupts localized virulence factor assembly sites in Enterococcus faecalis
Abstract
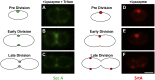
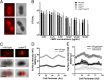
Virulence factor secretion and assembly occurs at spatially restricted foci in some Gram-positive bacteria. Given the essentiality of the general secretion pathway in bacteria and the contribution of virulence factors to disease progression, the foci that coordinate these processes are attractive antimicrobial targets. In this study, we show in Enterococcus faecalis that SecA and Sortase A, required for the attachment of virulence factors to the cell wall, localize to discrete domains near the septum or nascent septal site as the bacteria proceed through the cell cycle. We also demonstrate that cationic human β-defensins interact with E. faecalis at discrete septal foci, and this exposure disrupts sites of localized secretion and sorting. Modification of anionic lipids by multiple peptide resistance factor, a protein that confers antimicrobial peptide resistance by electrostatic repulsion, renders E. faecalis more resistant to killing by defensins and less susceptible to focal targeting by the cationic antimicrobial peptides. These data suggest a paradigm in which focal targeting by antimicrobial peptides is linked to their killing efficiency and to disruption of virulence factor assembly.
Keywords: MprF; focal localization; immunofluorescence; microscopy.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Comment in
-
Dual defensin strategy for targeting Enterococcus faecalis.Proc Natl Acad Sci U S A. 2013 Dec 10;110(50):19980-1. doi: 10.1073/pnas.1319939110. Epub 2013 Nov 27. Proc Natl Acad Sci U S A. 2013. PMID: 24284170 Free PMC article. No abstract available.
References
-
- Buist G, Ridder AN, Kok J, Kuipers OP. Different subcellular locations of secretome components of Gram-positive bacteria. Microbiology. 2006;152(Pt 10):2867–2874. - PubMed
-
- Rothfield L, Taghbalout A, Shih YL. Spatial control of bacterial division-site placement. Nat Rev Microbiol. 2005;3(12):959–968. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

