Selective G-quadruplex DNA recognition by a new class of designed cyanines
- PMID: 24192912
- PMCID: PMC4457452
- DOI: 10.3390/molecules181113588
Selective G-quadruplex DNA recognition by a new class of designed cyanines
Abstract
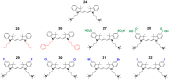
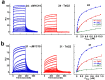
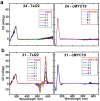
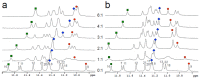
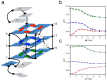
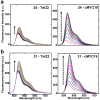
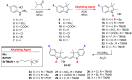
A variety of cyanines provide versatile and sensitive agents acting as DNA stains and sensors and have been structurally modified to bind in the DNA minor groove in a sequence dependent manner. Similarly, we are developing a new set of cyanines that have been designed to achieve highly selective binding to DNA G-quadruplexes with much weaker binding to DNA duplexes. A systematic set of structurally analogous trimethine cyanines has been synthesized and evaluated for quadruplex targeting. The results reveal that elevated quadruplex binding and specificity are highly sensitive to the polymethine chain length, heterocyclic structure and intrinsic charge of the compound. Biophysical experiments show that the compounds display significant selectivity for quadruplex binding with a higher preference for parallel stranded quadruplexes, such as cMYC. NMR studies revealed the primary binding through an end-stacking mode and SPR studies showed the strongest compounds have primary KD values below 100 nM that are nearly 100-fold weaker for duplexes. The high selectivity of these newly designed trimethine cyanines for quadruplexes as well as their ability to discriminate between different quadruplexes are extremely promising features to develop them as novel probes for targeting quadruplexes in vivo.
Figures
Similar articles
-
Second Generation G-Quadruplex Stabilizing Trimethine Cyanines.Bioconjug Chem. 2019 Oct 16;30(10):2647-2663. doi: 10.1021/acs.bioconjchem.9b00571. Epub 2019 Sep 26. Bioconjug Chem. 2019. PMID: 31518105 Free PMC article.
-
A dual-site simultaneous binding mode in the interaction between parallel-stranded G-quadruplex [d(TGGGGT)]4 and cyanine dye 2,2'-diethyl-9-methyl-selenacarbocyanine bromide.Nucleic Acids Res. 2013 Feb 1;41(4):2709-22. doi: 10.1093/nar/gks1328. Epub 2012 Dec 28. Nucleic Acids Res. 2013. PMID: 23275573 Free PMC article.
-
Halogenated pentamethine cyanine dyes exhibiting high fidelity for G-quadruplex DNA.Bioorg Med Chem. 2012 Dec 15;20(24):7002-11. doi: 10.1016/j.bmc.2012.10.008. Epub 2012 Oct 16. Bioorg Med Chem. 2012. PMID: 23127491 Free PMC article.
-
Interaction of G-quadruplexes with nonintercalating duplex-DNA minor groove binding ligands.Bioconjug Chem. 2011 Dec 21;22(12):2355-68. doi: 10.1021/bc200268a. Epub 2011 Nov 10. Bioconjug Chem. 2011. PMID: 22074555 Review.
-
NMR spectroscopy of G-quadruplexes.Methods. 2012 May;57(1):11-24. doi: 10.1016/j.ymeth.2012.05.003. Epub 2012 May 24. Methods. 2012. PMID: 22633887 Review.
Cited by
-
Second Generation G-Quadruplex Stabilizing Trimethine Cyanines.Bioconjug Chem. 2019 Oct 16;30(10):2647-2663. doi: 10.1021/acs.bioconjchem.9b00571. Epub 2019 Sep 26. Bioconjug Chem. 2019. PMID: 31518105 Free PMC article.
-
Exploration of cyanine compounds as selective inhibitors of protein arginine methyltransferases: synthesis and biological evaluation.J Med Chem. 2015 Feb 12;58(3):1228-43. doi: 10.1021/jm501452j. Epub 2015 Jan 21. J Med Chem. 2015. PMID: 25559100 Free PMC article.
-
Synthesis and Optical Properties of Pentamethine Cyanine Dyes With Carboxylic Acid Moieties.Anal Chem Insights. 2017 May 30;12:1177390117711938. doi: 10.1177/1177390117711938. eCollection 2017. Anal Chem Insights. 2017. PMID: 28607539 Free PMC article.
-
The Role of Small Molecules Containing Fluorine Atoms in Medicine and Imaging Applications.Pharmaceuticals (Basel). 2024 Feb 22;17(3):281. doi: 10.3390/ph17030281. Pharmaceuticals (Basel). 2024. PMID: 38543068 Free PMC article. Review.
-
Benz[c,d]indolium-containing Monomethine Cyanine Dyes: Synthesis and Photophysical Properties.Molecules. 2015 Dec 24;21(1):E23. doi: 10.3390/molecules21010023. Molecules. 2015. PMID: 26712725 Free PMC article.
References
-
- Neidle S., Balasubramanian S. Quadruplex Nucleic Acids. RSC Publishers; Cambridge, UK: 2006. pp. 301–315.
-
- Schaffitzel C., Postberg J., Paeschke K., Lipps H. Probing Telomeric G-Quadruplex DNA Structures in Cells with in Vitro Generated Single-Chain Antibody Fragments. In: Baumann P., editor. G-Quadruplex DNA. Volume 608. Humana Press; New York, NY, USA: 2010. pp. 159–181. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

