Regulation of epithelial plasticity by miR-424 and miR-200 in a new prostate cancer metastasis model
- PMID: 24193225
- PMCID: PMC3818652
- DOI: 10.1038/srep03151
Regulation of epithelial plasticity by miR-424 and miR-200 in a new prostate cancer metastasis model
Abstract
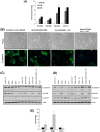
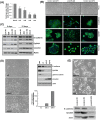
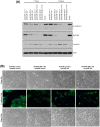
Using an in vivo cycling strategy, we selected metastatic cancer cells from the lymph nodes (LN) of mice bearing orthotopic DU145 human prostate tumors. Repeated rounds of metastatic selection (LN1-LN4) progressively increased the epithelial phenotype, resulting in a new model of tumor cell mesenchymal-epithelial transition (MET). DU145-LN4 showed increased cell-cell adhesions, higher expression of multiple epithelial markers, such as E-cadherin, EpCAM and cytokeratin 18, and reduced expression of mesenchymal markers such as vimentin. The MET in DU145-LN4 cells was accompanied by increased expression of the miR-200 family, and antimiRs to miR-200c and miR-141 induced an EMT. MET also correlated with the loss of miR-424. Ectopic transient and stable miR-424 expression induced EMT, with reduced epithelial marker expression and increased cell scattering. Our model provides evidence for spontaneous MET in vivo. We show that this cellular plasticity can be mediated through the combined action of miR-424 and the miR-200 family.
Figures




References
-
- Gupta G. P. & Massague J. Cancer metastasis: building a framework. Cell 127, 679–695 (2006). - PubMed
-
- Tarin D., Thompson E. W. & Newgreen D. F. The fallacy of epithelial mesenchymal transition in neoplasia. Cancer Res 65, 5996–6000; discussion 6000-5991; (2005). - PubMed
-
- Thompson E. W., Newgreen D. F. & Tarin D. Carcinoma invasion and metastasis: a role for epithelial-mesenchymal transition? Cancer Res 65, 5991–5995 discussion 5995 (2005). - PubMed
-
- Garber K. Epithelial-to-mesenchymal transition is important to metastasis, but questions remain. J Natl Cancer Inst 100, 232–233 239 (2008). - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

