Lactadherin inhibits secretory phospholipase A2 activity on pre-apoptotic leukemia cells
- PMID: 24194865
- PMCID: PMC3806724
- DOI: 10.1371/journal.pone.0077143
Lactadherin inhibits secretory phospholipase A2 activity on pre-apoptotic leukemia cells
Abstract
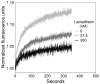
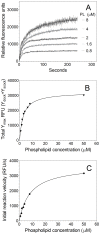
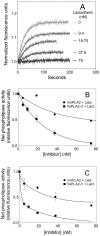
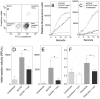
Secretory phospholipase A2 (sPLA2) is a critical component of insect and snake venoms and is secreted by mammalian leukocytes during inflammation. Elevated secretory PLA2 concentrations are associated with autoimmune diseases and septic shock. Many sPLA2's do not bind to plasma membranes of quiescent cells but bind and digest phospholipids on the membranes of stimulated or apoptotic cells. The capacity of these phospholipases to digest membranes of stimulated or apoptotic cells correlates to the exposure of phosphatidylserine. In the present study, the ability of the phosphatidyl-L-serine-binding protein, lactadherin to inhibit phospholipase enzyme activity has been assessed. Inhibition of human secretory phospholipase A2-V on phospholipid vesicles exceeded 90%, whereas inhibition of Naja mossambica sPLA2 plateaued at 50-60%. Lactadherin inhibited 45% of activity of Naja mossambica sPLA2 and >70% of human secretory phospholipase A2-V on the membranes of human NB4 leukemia cells treated with calcium ionophore A23187. The data indicate that lactadherin may decrease inflammation by inhibiting sPLA2.
Conflict of interest statement
Figures
Similar articles
-
Inhibition of secreted phospholipases A2 by annexin V. Competition for anionic phospholipid interfaces allows an assessment of the relative interfacial affinities of secreted phospholipases A2.Biochim Biophys Acta. 1998 Apr 22;1391(3):367-76. doi: 10.1016/s0005-2760(98)00026-5. Biochim Biophys Acta. 1998. PMID: 9555096
-
Sphingomyelin Regulates the Activity of Secretory Phospholipase A2 in the Plasma Membrane.J Cell Biochem. 2015 Sep;116(9):1898-907. doi: 10.1002/jcb.25145. J Cell Biochem. 2015. PMID: 25716287
-
Lactadherin inhibits enzyme complexes of blood coagulation by competing for phospholipid-binding sites.Blood. 2003 Apr 1;101(7):2628-36. doi: 10.1182/blood-2002-07-1951. Epub 2002 Nov 27. Blood. 2003. PMID: 12517809
-
Modulation of immunity by the secreted phospholipase A2 family.Immunol Rev. 2023 Aug;317(1):42-70. doi: 10.1111/imr.13205. Epub 2023 Apr 10. Immunol Rev. 2023. PMID: 37035998 Review.
-
Roles of secreted phospholipase A2 group IIA in inflammation and host defense.Biochim Biophys Acta Mol Cell Biol Lipids. 2019 Jun;1864(6):789-802. doi: 10.1016/j.bbalip.2018.08.017. Epub 2018 Sep 13. Biochim Biophys Acta Mol Cell Biol Lipids. 2019. PMID: 30905346 Review.
Cited by
-
Lactadherin orthologs inhibit migration of human, porcine and murine intestinal epithelial cells.Food Sci Nutr. 2017 May 15;5(4):934-942. doi: 10.1002/fsn3.479. eCollection 2017 Jul. Food Sci Nutr. 2017. PMID: 28748083 Free PMC article.
-
The Potential Clinical Relevance of Procoagulant Microparticles as Biomarkers of Blood Coagulation in Breast Cancer: A Systematic Review.Asian Pac J Cancer Prev. 2025 Jan 1;26(1):23-32. doi: 10.31557/APJCP.2025.26.1.23. Asian Pac J Cancer Prev. 2025. PMID: 39873982 Free PMC article.
-
Dietary proteins and angiogenesis.Nutrients. 2014 Jan 17;6(1):371-81. doi: 10.3390/nu6010371. Nutrients. 2014. PMID: 24445377 Free PMC article. Review.
References
-
- Kuipers OP, van den Bergh CJ, Verheij HM, de Haas GH (1990) Probing the mechanism of pancreatic phospholipase A2 with the aid of recombinant DNA techniques. Adv Exp Med Biol 279: 65–84. - PubMed
-
- Jain MK, Rogers J, Jahagirdar DV, Marecek JF, Ramirez F (1986) Kinetics of interfacial catalysis by phospholipase A2 in intravesicle scooting mode, and heterofusion of anionic and zwitterionic vesicles. Biochimica et Biophysica Acta (BBA) - Biomembranes 860: 435–447 10.1016/0005-2736(86)90541-9 - DOI - PubMed
-
- Hara S, Kudo I, Chang HW, Matsuta K, Miyamoto T, et al. (1989) Purification and characterization of extracellular phospholipase A2 from human synovial fluid in rheumatoid arthritis. J Biochem 105: 395–399. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

