The type II deiodinase is retrotranslocated to the cytoplasm and proteasomes via p97/Atx3 complex
- PMID: 24196352
- PMCID: PMC3857201
- DOI: 10.1210/me.2013-1281
The type II deiodinase is retrotranslocated to the cytoplasm and proteasomes via p97/Atx3 complex
Abstract
The type II iodothyronine deiodinase (D2) is a type I endoplasmic reticulum (ER)-resident thioredoxin fold-containing selenoprotein that activates thyroid hormone. D2 is inactivated by ER-associated ubiquitination and can be reactivated by two ubiquitin-specific peptidase-class D2-interacting deubiquitinases (DUBs). Here, we used D2-expressing cell models to define that D2 ubiquitination (UbD2) occurs via K48-linked ubiquitin chains and that exposure to its natural substrate, T4, accelerates UbD2 formation and retrotranslocation to the cytoplasm via interaction with the p97-ATPase complex. D2 retrotranslocation also includes deubiquitination by the p97-associated DUB Ataxin-3 (Atx3). Inhibiting Atx3 with eeyarestatin-I did not affect D2:p97 binding but decreased UbD2 retrotranslocation and caused ER accumulation of high-molecular weight UbD2 bands possibly by interfering with the D2-ubiquitin-specific peptidases binding. Once in the cytosol, D2 is delivered to the proteasomes as evidenced by coprecipitation with 19S proteasome subunit S5a and increased colocalization with the 20S proteasome. We conclude that interaction between UbD2 and p97/Atx3 mediates retranslocation of UbD2 to the cytoplasm for terminal degradation in the proteasomes, a pathway that is accelerated by exposure to T4.
Figures

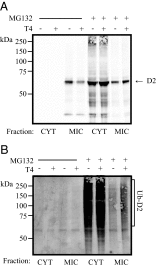
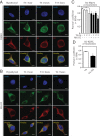
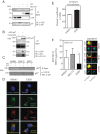
References
-
- Ciehanover A, Hod Y, Hershko A. A heat-stable polypeptide component of an ATP-dependent proteolytic system from reticulocytes. Biochem Biophys Res Commun. 1978;81:1100–1105 - PubMed
-
- Komander D, Rape M. The ubiquitin code. Annu Rev Biochem. 2012;81:203–229 - PubMed
-
- Mukhopadhyay D, Riezman H. Proteasome-independent functions of ubiquitin in endocytosis and signaling. Science. 2007;315:201–205 - PubMed
-
- Chau V, Tobias JW, Bachmair A, et al. A multiubiquitin chain is confined to specific lysine in a targeted short-lived protein. Science. 1989;243:1576–1583 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

