Src and CXCR4 are involved in the invasiveness of breast cancer cells with acquired resistance to lapatinib
- PMID: 24200972
- PMCID: PMC3925726
- DOI: 10.4161/cc.26899
Src and CXCR4 are involved in the invasiveness of breast cancer cells with acquired resistance to lapatinib
Abstract
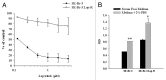
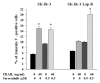
Lapatinib is a dual EGFR and ErbB-2 tyrosine kinase inhibitor that has significantly improved the clinical outcome of ErbB-2-overexpressing breast cancer patients. However, patients inexorably develop mechanisms of resistance that limit the efficacy of the drug. In order to identify potential targets for therapeutic intervention in lapatinib-resistant patients, we isolated, from ErbB-2-overexpressing SK-Br-3 breast cancer cells, the SK-Br-3 Lap-R-resistant subclone, which is able to routinely grow in 1 µM lapatinib. Resistant cells have a more aggressive phenotype compared with parental cells, as they show a higher ability to invade through a matrigel-coated membrane. Lapatinib-resistant cells have an increased Src kinase activity and persistent levels of activation of ERK1/2 and AKT compared with parental cells. Treatment with the Src inhibitor saracatinib in combination with lapatinib reduces AKT and ERK1/2 phosphorylation and restores the sensitivity of resistant cells to lapatinib. SK-Br-3 Lap-R cells also show levels of expression of CXCR4 that are higher compared with parental cells and are not affected by Src inhibition. Treatment with saracatinib or a specific CXCR4 antibody reduces the invasive ability of SK-Br-3 Lap-R cells, with the two drugs showing cooperative effects. Finally, blockade of Src signaling significantly increases TRAIL-induced cell death in SK-Br-3 Lap-R cells. Taken together, our results demonstrate that breast cancer cells with acquired resistance to lapatinib have a more aggressive phenotype compared with their parental counterpart, and that Src signaling and CXCR4 play an important role in this phenomenon, thus representing potential targets for therapeutic intervention in lapatinib-resistant breast cancer patients.
Keywords: CXCR4; ErbB-2; Src kinase; breast cancer; lapatinib; resistance; saracatinib.
Figures




Similar articles
-
Epidermal growth factor-receptor activation modulates Src-dependent resistance to lapatinib in breast cancer models.Breast Cancer Res. 2014 May 5;16(3):R45. doi: 10.1186/bcr3650. Breast Cancer Res. 2014. PMID: 24887236 Free PMC article.
-
Combined effects of lapatinib and bortezomib in human epidermal receptor 2 (HER2)-overexpressing breast cancer cells and activity of bortezomib against lapatinib-resistant breast cancer cells.Cancer Sci. 2010 Oct;101(10):2220-6. doi: 10.1111/j.1349-7006.2010.01662.x. Epub 2010 Aug 5. Cancer Sci. 2010. PMID: 20701607 Free PMC article.
-
Yes1 signaling mediates the resistance to Trastuzumab/Lap atinib in breast cancer.PLoS One. 2017 Feb 3;12(2):e0171356. doi: 10.1371/journal.pone.0171356. eCollection 2017. PLoS One. 2017. PMID: 28158234 Free PMC article.
-
Antitumor activity of saracatinib (AZD0530), a c-Src/Abl kinase inhibitor, alone or in combination with chemotherapeutic agents in gastric cancer.Mol Cancer Ther. 2013 Jan;12(1):16-26. doi: 10.1158/1535-7163.MCT-12-0109. Epub 2012 Nov 9. Mol Cancer Ther. 2013. PMID: 23144237
-
Phosphoproteomic mass spectrometry profiling links Src family kinases to escape from HER2 tyrosine kinase inhibition.Oncogene. 2011 Oct 6;30(40):4163-74. doi: 10.1038/onc.2011.130. Epub 2011 Apr 18. Oncogene. 2011. PMID: 21499296 Free PMC article.
Cited by
-
Identification and characterization of biomarkers and their functions for Lapatinib-resistant breast cancer.Med Oncol. 2017 May;34(5):89. doi: 10.1007/s12032-017-0953-y. Epub 2017 Apr 9. Med Oncol. 2017. PMID: 28393315
-
SRC Kinase in Glioblastoma News from an Old Acquaintance.Cancers (Basel). 2020 Jun 12;12(6):1558. doi: 10.3390/cancers12061558. Cancers (Basel). 2020. PMID: 32545574 Free PMC article. Review.
-
Therapeutic targeting of ERBB2 in breast cancer: understanding resistance in the laboratory and combating it in the clinic.J Mol Med (Berl). 2014 Jul;92(7):681-95. doi: 10.1007/s00109-014-1169-7. Epub 2014 May 28. J Mol Med (Berl). 2014. PMID: 24861025 Review.
-
Role of Chemokines in the Biology of Cholangiocarcinoma.Cancers (Basel). 2020 Aug 7;12(8):2215. doi: 10.3390/cancers12082215. Cancers (Basel). 2020. PMID: 32784743 Free PMC article. Review.
-
The Role of the CXCL12/CXCR4/CXCR7 Chemokine Axis in Cancer.Front Pharmacol. 2020 Dec 8;11:574667. doi: 10.3389/fphar.2020.574667. eCollection 2020. Front Pharmacol. 2020. PMID: 33363463 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous
