Cost-effectiveness of modified-release prednisone in the treatment of moderate to severe rheumatoid arthritis with morning stiffness based on directly elicited public preference values
- PMID: 24204166
- PMCID: PMC3816994
- DOI: 10.2147/CEOR.S47867
Cost-effectiveness of modified-release prednisone in the treatment of moderate to severe rheumatoid arthritis with morning stiffness based on directly elicited public preference values
Abstract
Background: Assessing the cost-effectiveness of treatments in rheumatoid arthritis (RA) is of growing importance due to the chronic nature of the disease, rising treatment costs, and budget-constrained health care systems. This analysis assesses the cost-effectiveness of modified-release (MR) prednisone compared with immediate-release (IR) prednisone for the treatment of morning stiffness due to RA.
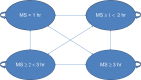
Methods: A health state transition model was used to categorize RA patients into four health states, defined by duration of morning stiffness. The model applied a 1-year time horizon and adopted a UK National Health Service (NHS) perspective. Health benefits were measured in quality-adjusted life years (QALYs) and the final output was the incremental cost-effectiveness ratio (ICER). Efficacy data were derived from the CAPRA-1 (Circadian Administration of Prednisone in Rheumatoid Arthritis) study, drug costs from the British National Formulary (BNF), and utility data from a direct elicitation time-trade-off (TTO) study in the general population. Sensitivity analyses were conducted.
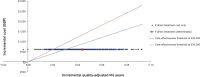
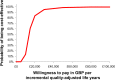
Results: Mean treatment costs per patient were higher for MR-prednisone (£649.70) than for IR-prednisone (£46.54) for the duration of the model. However, the model generated an incremental QALY of 0.044 in favor of MR-prednisone which resulted in an ICER of £13,577. Deterministic sensitivity analyses did not lead to significant changes in the ICER. Probabilistic sensitivity analysis reported that MR-prednisone had an 84% probability of being cost-effective at a willingness-to-pay threshold of £30,000 per QALY. The model only considers drug costs and there was a lack of comparative long-term data for IR-prednisone. Furthermore, utility benefits were not captured in the clinical setting.
Conclusion: This analysis demonstrates that, based on the CAPRA-1 trial and directly elicited public preference values, MR-prednisone is a cost-effective treatment option when compared with IR-prednisone for RA patients with morning stiffness over one year, according to commonly applied UK thresholds (£20,000-£30,000 per QALY). Further research into the costs of morning stiffness in RA is required.
Keywords: cost utility analysis; cost-effectiveness analysis; modified-release prednisone; morning stiffness; quality of life; rheumatoid arthritis.
Figures
References
-
- Lundkvist J, Kastang F, Kobelt G. The burden of rhematoid arthritis and access to treatments: health burden and costs. Eur J Health Econ. 2008;8(2):49–60. - PubMed
-
- Grassi W, De AR, Lamanna G, Cervini C. The clinical features of rheumatoid arthritis. Eur J Radiol. 1998;27(1):18–24. - PubMed
-
- Adams R, Walsh C, Veale D, Bresnihan B, FitzGerald O, Barry M. Understanding the relationship between the EQ-5D, SF-6D, HAQ and disease activity in inflammatory arthritis. Pharmacoeconomics. 2010;28(6):477–487. - PubMed
-
- Cutolo M. How should morning function in rheumatoid arthritis be assessed? Bibliographic study of current assessment. Scand J Rheumatol Suppl. 2011;125:17–22. - PubMed
-
- Sierakowski S, Cutolo M. Morning symptoms in rheumatoid arthritis: a defining characteristic and marker of active disease. Scand J Rheumatol Suppl. 2011;125:1–5. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

