Potassium buffering in the neurovascular unit: models and sensitivity analysis
- PMID: 24209849
- PMCID: PMC3824545
- DOI: 10.1016/j.bpj.2013.09.012
Potassium buffering in the neurovascular unit: models and sensitivity analysis
Abstract
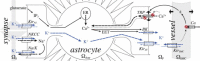
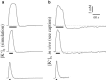
Astrocytes are critical regulators of neural and neurovascular network communication. Potassium transport is a central mechanism behind their many functions. Astrocytes encircle synapses with their distal processes, which express two potassium pumps (Na-K and NKCC) and an inward rectifying potassium channel (Kir), whereas the vessel-adjacent endfeet express Kir and BK potassium channels. We provide a detailed model of potassium flow throughout the neurovascular unit (synaptic region, astrocytes, and arteriole) for the cortex of the young brain. Our model reproduces several phenomena observed experimentally: functional hyperemia, in which neural activity triggers astrocytic potassium release at the perivascular endfoot, inducing arteriole dilation; K(+) undershoot in the synaptic space after periods of neural activity; neurally induced astrocyte hyperpolarization during Kir blockade. Our results suggest that the dynamics of the vascular response during functional hyperemia are governed by astrocytic Kir for the fast onset and astrocytic BK for maintaining dilation. The model supports the hypothesis that K(+) undershoot is caused by excessive astrocytic uptake through Na-K and NKCC pumps, whereas the effect is balanced by Kir. We address parametric uncertainty using high-dimensional stochastic sensitivity analysis and identify possible model limitations.
Copyright © 2013 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Figures



Similar articles
-
Mechanisms of astrocytic K(+) clearance and swelling under high extracellular K(+) concentrations.J Physiol Sci. 2016 Mar;66(2):127-42. doi: 10.1007/s12576-015-0404-5. Epub 2015 Oct 27. J Physiol Sci. 2016. PMID: 26507417 Free PMC article.
-
Differential role of KIR channel and Na(+)/K(+)-pump in the regulation of extracellular K(+) in rat hippocampus.J Neurophysiol. 2002 Jan;87(1):87-102. doi: 10.1152/jn.00240.2001. J Neurophysiol. 2002. PMID: 11784732
-
Inversion of neurovascular coupling by subarachnoid blood depends on large-conductance Ca2+-activated K+ (BK) channels.Proc Natl Acad Sci U S A. 2012 May 22;109(21):E1387-95. doi: 10.1073/pnas.1121359109. Epub 2012 Apr 30. Proc Natl Acad Sci U S A. 2012. PMID: 22547803 Free PMC article.
-
Emerging Roles of Astrocyte Kir4.1 Channels in the Pathogenesis and Treatment of Brain Diseases.Int J Mol Sci. 2021 Sep 23;22(19):10236. doi: 10.3390/ijms221910236. Int J Mol Sci. 2021. PMID: 34638578 Free PMC article. Review.
-
Vascular inward rectifier K+ channels as external K+ sensors in the control of cerebral blood flow.Microcirculation. 2015 Apr;22(3):183-96. doi: 10.1111/micc.12190. Microcirculation. 2015. PMID: 25641345 Free PMC article. Review.
Cited by
-
Channel-mediated lactate release by K⁺-stimulated astrocytes.J Neurosci. 2015 Mar 11;35(10):4168-78. doi: 10.1523/JNEUROSCI.5036-14.2015. J Neurosci. 2015. PMID: 25762664 Free PMC article.
-
The Role of Cell Volume in the Dynamics of Seizure, Spreading Depression, and Anoxic Depolarization.PLoS Comput Biol. 2015 Aug 14;11(8):e1004414. doi: 10.1371/journal.pcbi.1004414. eCollection 2015 Aug. PLoS Comput Biol. 2015. PMID: 26273829 Free PMC article.
-
Ion channel networks in the control of cerebral blood flow.J Cereb Blood Flow Metab. 2016 Mar;36(3):492-512. doi: 10.1177/0271678X15616138. Epub 2015 Nov 9. J Cereb Blood Flow Metab. 2016. PMID: 26661232 Free PMC article. Review.
-
The role of astrocytic calcium and TRPV4 channels in neurovascular coupling.J Comput Neurosci. 2018 Feb;44(1):97-114. doi: 10.1007/s10827-017-0671-7. Epub 2017 Nov 20. J Comput Neurosci. 2018. PMID: 29152668
-
A Computational Study of Astrocytic GABA Release at the Glutamatergic Synapse: EAAT-2 and GAT-3 Coupled Dynamics.Front Cell Neurosci. 2021 Jul 12;15:682460. doi: 10.3389/fncel.2021.682460. eCollection 2021. Front Cell Neurosci. 2021. PMID: 34322000 Free PMC article.
References
-
- Witthoft A., Em Karniadakis G. A bidirectional model for communication in the neurovascular unit. J. Theor. Biol. 2012;311:80–93. - PubMed
-
- Ishii M., Fujita A., Kurachi Y. Differential expression and distribution of Kir5.1 and Kir4.1 inwardly rectifying K+ channels in retina. Am. J. Physiol. Cell Physiol. 2003;285:C260–C267. - PubMed
-
- Kofuji P., Biedermann B., Reichenbach A. Kir potassium channel subunit expression in retinal glial cells: implications for spatial potassium buffering. Glia. 2002;39:292–303. - PubMed
-
- Neusch C., Papadopoulos N., Hülsmann S. Lack of the Kir4.1 channel subunit abolishes K+ buffering properties of astrocytes in the ventral respiratory group: impact on extracellular K+ regulation. J. Neurophysiol. 2006;95:1843–1852. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

