Genome-wide consequences of deleting any single gene
- PMID: 24211263
- PMCID: PMC3975072
- DOI: 10.1016/j.molcel.2013.09.026
Genome-wide consequences of deleting any single gene
Abstract
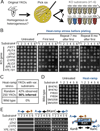
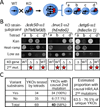
Loss or duplication of chromosome segments can lead to further genomic changes associated with cancer. However, it is not known whether only a select subset of genes is responsible for driving further changes. To determine whether perturbation of any given gene in a genome suffices to drive subsequent genetic changes, we analyzed the yeast knockout collection for secondary mutations of functional consequence. Unlike wild-type, most gene knockout strains were found to have one additional mutant gene affecting nutrient responses and/or heat-stress-induced cell death. Moreover, independent knockouts of the same gene often evolved mutations in the same secondary gene. Genome sequencing identified acquired mutations in several human tumor suppressor homologs. Thus, mutation of any single gene may cause a genomic imbalance, with consequences sufficient to drive adaptive genetic changes. This complicates genetic analyses but is a logical consequence of losing a functional unit originally acquired under pressure during evolution.
Copyright © 2013 Elsevier Inc. All rights reserved.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures





Comment in
-
Genome evolution in yeast reveals connections between rare mutations in human cancers.Microb Cell. 2014 Jun 2;1(6):206-209. doi: 10.15698/mic2014.06.153. Microb Cell. 2014. PMID: 28357245 Free PMC article.
References
-
- Barrick JE, Yu DS, Yoon SH, Jeong H, Oh TK, Schneider D, Lenski RE, Kim JF. Genome evolution and adaptation in a long-term experiment with Escherichia coli. Nature. 2009;461:1243–1247. - PubMed
Publication types
MeSH terms
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

