Genetic deletion of afadin causes hydrocephalus by destruction of adherens junctions in radial glial and ependymal cells in the midbrain
- PMID: 24236178
- PMCID: PMC3827428
- DOI: 10.1371/journal.pone.0080356
Genetic deletion of afadin causes hydrocephalus by destruction of adherens junctions in radial glial and ependymal cells in the midbrain
Abstract
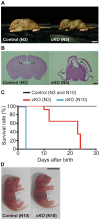
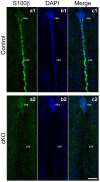
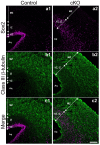
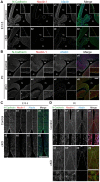
Adherens junctions (AJs) play a role in mechanically connecting adjacent cells to maintain tissue structure, particularly in epithelial cells. The major cell-cell adhesion molecules at AJs are cadherins and nectins. Afadin binds to both nectins and α-catenin and recruits the cadherin-β-catenin complex to the nectin-based cell-cell adhesion site to form AJs. To explore the role of afadin in radial glial and ependymal cells in the brain, we generated mice carrying a nestin-Cre-mediated conditional knockout (cKO) of the afadin gene. Newborn afadin-cKO mice developed hydrocephalus and died neonatally. The afadin-cKO brain displayed enlarged lateral ventricles and cerebral aqueduct, resulting from stenosis of the caudal end of the cerebral aqueduct and obliteration of the ventral part of the third ventricle. Afadin deficiency further caused the loss of ependymal cells from the ventricular and aqueductal surfaces. During development, radial glial cells, which terminally differentiate into ependymal cells, scattered from the ventricular zone and were replaced by neurons that eventually covered the ventricular and aqueductal surfaces of the afadin-cKO midbrain. Moreover, the denuded ependymal cells were only occasionally observed in the third ventricle and the cerebral aqueduct of the afadin-cKO midbrain. Afadin was co-localized with nectin-1 and N-cadherin at AJs of radial glial and ependymal cells in the control midbrain, but these proteins were not concentrated at AJs in the afadin-cKO midbrain. Thus, the defects in the afadin-cKO midbrain most likely resulted from the destruction of AJs, because AJs in the midbrain were already established before afadin was genetically deleted. These results indicate that afadin is essential for the maintenance of AJs in radial glial and ependymal cells in the midbrain and is required for normal morphogenesis of the cerebral aqueduct and ventral third ventricle in the midbrain.
Conflict of interest statement
Figures


Similar articles
-
Involvement of LMO7 in the association of two cell-cell adhesion molecules, nectin and E-cadherin, through afadin and alpha-actinin in epithelial cells.J Biol Chem. 2004 Jul 23;279(30):31365-73. doi: 10.1074/jbc.M401957200. Epub 2004 May 12. J Biol Chem. 2004. PMID: 15140894
-
Cooperative roles of Par-3 and afadin in the formation of adherens and tight junctions.J Cell Sci. 2007 Jul 15;120(Pt 14):2352-65. doi: 10.1242/jcs.03470. J Cell Sci. 2007. PMID: 17606991
-
Regulation of the assembly and adhesion activity of E-cadherin by nectin and afadin for the formation of adherens junctions in Madin-Darby canine kidney cells.J Biol Chem. 2006 Feb 24;281(8):5288-99. doi: 10.1074/jbc.M510070200. Epub 2005 Dec 18. J Biol Chem. 2006. PMID: 16361708
-
Adherent junctions: Physiology, role in hydrocephalus and potential therapeutic targets.IBRO Neurosci Rep. 2025 Feb 6;18:283-292. doi: 10.1016/j.ibneur.2025.02.003. eCollection 2025 Jun. IBRO Neurosci Rep. 2025. PMID: 39995568 Free PMC article. Review.
-
Adherens Junctions: Guardians of Cortical Development.Front Cell Dev Biol. 2020 Jan 28;8:6. doi: 10.3389/fcell.2020.00006. eCollection 2020. Front Cell Dev Biol. 2020. PMID: 32117958 Free PMC article. Review.
Cited by
-
SAX-7/L1CAM acts with the adherens junction proteins MAGI-1, HMR-1/Cadherin, and AFD-1/Afadin to promote glial-mediated dendrite extension.bioRxiv [Preprint]. 2024 Jan 11:2024.01.11.575259. doi: 10.1101/2024.01.11.575259. bioRxiv. 2024. PMID: 38260503 Free PMC article. Preprint.
-
Proliferative defects and formation of a double cortex in mice lacking Mltt4 and Cdh2 in the dorsal telencephalon.J Neurosci. 2014 Aug 6;34(32):10475-87. doi: 10.1523/JNEUROSCI.1793-14.2014. J Neurosci. 2014. PMID: 25100583 Free PMC article.
-
IIIG9 inhibition in adult ependymal cells changes adherens junctions structure and induces cellular detachment.Sci Rep. 2021 Sep 17;11(1):18537. doi: 10.1038/s41598-021-97948-3. Sci Rep. 2021. PMID: 34535732 Free PMC article.
-
Afadin requirement for cytokine expressions in keratinocytes during chemically induced inflammation in mice.Genes Cells. 2014 Nov;19(11):842-52. doi: 10.1111/gtc.12184. Epub 2014 Oct 9. Genes Cells. 2014. PMID: 25297509 Free PMC article.
-
Cumulative Damage: Cell Death in Posthemorrhagic Hydrocephalus of Prematurity.Cells. 2021 Jul 28;10(8):1911. doi: 10.3390/cells10081911. Cells. 2021. PMID: 34440681 Free PMC article. Review.
References
-
- Del Bigio MR (2010) Ependymal cells: biology and pathology. Acta Neuropathol 119: 55–73. - PubMed
-
- Götz M, Huttner WB (2005) The cell biology of neurogenesis. Nat Rev Mol Cell Biol 6: 777–788. - PubMed
-
- Gumbiner BM (1996) Cell adhesion: the molecular basis of tissue architecture and morphogenesis. Cell 84: 345–357. - PubMed
-
- Schneeberger EE, Lynch RD (1992) Structure, function, and regulation of cellular tight junctions. Am J Physiol 262: L647–661. - PubMed
-
- González-Mariscal L, Betanzos A, Nava P, Jaramillo BE (2003) Tight junction proteins. Prog Biophys Mol Biol 81: 1–44. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

