Human cytomegalovirus tegument protein pUL83 inhibits IFI16-mediated DNA sensing for immune evasion
- PMID: 24237704
- PMCID: PMC3876934
- DOI: 10.1016/j.chom.2013.10.007
Human cytomegalovirus tegument protein pUL83 inhibits IFI16-mediated DNA sensing for immune evasion
Abstract
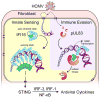
Nuclear sensing of viral DNA has emerged as an essential step in innate immune responses against herpesviruses. Here, we provide mechanistic insight into host recognition of human cytomegalovirus (HCMV) and subsequent immune evasion by this prominent DNA virus. We establish that the interferon-inducible protein IFI16 acts as a nuclear DNA sensor following HCMV infection, binding viral DNA and triggering expression of antiviral cytokines via the STING-TBK1-IRF3 signaling pathway. The HCMV tegument protein pUL83 inhibits this response by interacting with the IFI16 pyrin domain, blocking its oligomerization upon DNA sensing and subsequent immune signals. pUL83 disrupts IFI16 by concerted action of its N- and C-terminal domains, in which an evolutionarily conserved N-terminal pyrin association domain (PAD) binds IFI16. Additionally, phosphorylation of the N-terminal domain modulates pUL83-mediated inhibition of pyrin aggregation. Collectively, our data elucidate the interplay between host DNA sensing and HCMV immune evasion, providing targets for restoring antiviral immunity.
Copyright © 2013 Elsevier Inc. All rights reserved.
Figures


References
-
- Arnon TI, Achdout H, Levi O, Markel G, Saleh N, Katz G, Gazit R, Gonen-Gross T, Hanna J, Nahari E, et al. Inhibition of the NKp30 activating receptor by pp65 of human cytomegalovirus. Nat Immunol. 2005;6:515–523. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

