The end of an old hypothesis: the pseudomonas signaling molecules 4-hydroxy-2-alkylquinolines derive from fatty acids, not 3-ketofatty acids
- PMID: 24239007
- PMCID: PMC3877684
- DOI: 10.1016/j.chembiol.2013.09.021
The end of an old hypothesis: the pseudomonas signaling molecules 4-hydroxy-2-alkylquinolines derive from fatty acids, not 3-ketofatty acids
Abstract
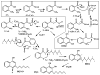
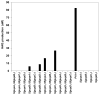
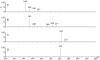
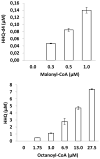
Groups of pathogenic bacteria use diffusible signals to regulate their virulence in a concerted manner. Pseudomonas aeruginosa uses 4-hydroxy-2-alkylquinolines (HAQs), including 4-hydroxy-2-heptylquinoline (HHQ) and 3,4-dihydroxy-2-heptylquinoline (PQS), as unique signals. We demonstrate that octanoic acid is directly incorporated into HHQ. This finding rules out the long-standing hypothesis that 3-ketofatty acids are the precursors of HAQs. We found that HAQ biosynthesis, which requires the PqsABCD enzymes, proceeds by a two-step pathway: (1) PqsD mediates the synthesis of 2-aminobenzoylacetate (2-ABA) from anthraniloyl-coenzyme A (CoA) and malonyl-CoA, then (2) the decarboxylating coupling of 2-ABA to an octanoate group linked to PqsC produces HHQ, the direct precursor of PQS. PqsB is tightly associated with PqsC and required for the second step. This finding uncovers promising targets for the development of specific antivirulence drugs to combat this opportunistic pathogen.
Copyright © 2013 Elsevier Ltd. All rights reserved.
Figures

References
-
- Bjarnsholt T, Tolker-Nielsen T, Hoiby N, Givskov M. Interference of Pseudomonas aeruginosa signalling and biofilm formation for infection control. Expert Rev Mol Med. 2010;12:e11. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

