Pseudomonas aeruginosa Cif protein enhances the ubiquitination and proteasomal degradation of the transporter associated with antigen processing (TAP) and reduces major histocompatibility complex (MHC) class I antigen presentation
- PMID: 24247241
- PMCID: PMC3879540
- DOI: 10.1074/jbc.M113.459271
Pseudomonas aeruginosa Cif protein enhances the ubiquitination and proteasomal degradation of the transporter associated with antigen processing (TAP) and reduces major histocompatibility complex (MHC) class I antigen presentation
Abstract
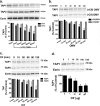
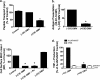
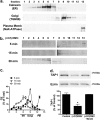
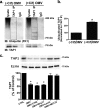
Cif (PA2934), a bacterial virulence factor secreted in outer membrane vesicles by Pseudomonas aeruginosa, increases the ubiquitination and lysosomal degradation of some, but not all, plasma membrane ATP-binding cassette transporters (ABC), including the cystic fibrosis transmembrane conductance regulator and P-glycoprotein. The goal of this study was to determine whether Cif enhances the ubiquitination and degradation of the transporter associated with antigen processing (TAP1 and TAP2), members of the ABC transporter family that play an essential role in antigen presentation and intracellular pathogen clearance. Cif selectively increased the amount of ubiquitinated TAP1 and increased its degradation in the proteasome of human airway epithelial cells. This effect of Cif was mediated by reducing USP10 deubiquitinating activity, resulting in increased polyubiquitination and proteasomal degradation of TAP1. The reduction in TAP1 abundance decreased peptide antigen translocation into the endoplasmic reticulum, an effect that resulted in reduced antigen available to MHC class I molecules for presentation at the plasma membrane of airway epithelial cells and recognition by CD8(+) T cells. Cif is the first bacterial factor identified that inhibits TAP function and MHC class I antigen presentation.
Keywords: ABC Transporter; Airway Epithelial Cell; Antigen Processing; Deubiquitination; Pseudomonas aeruginosa; TAP; Ubiquitination; Viral-Bacterial Co-infection.
Figures

Similar articles
-
Restoration of the expression of transports associated with antigen processing in human malignant melanoma increases tumor-specific immunity.J Invest Dermatol. 2008 Aug;128(8):1991-6. doi: 10.1038/jid.2008.10. Epub 2008 Apr 3. J Invest Dermatol. 2008. PMID: 18385764
-
Exploring the minimal functional unit of the transporter associated with antigen processing.FEBS Lett. 2005 Aug 15;579(20):4413-6. doi: 10.1016/j.febslet.2005.07.006. FEBS Lett. 2005. PMID: 16061226
-
Peptide-bound major histocompatibility complex class I molecules associate with tapasin before dissociation from transporter associated with antigen processing.J Biol Chem. 1999 Mar 26;274(13):8649-54. doi: 10.1074/jbc.274.13.8649. J Biol Chem. 1999. PMID: 10085102
-
Involvement of autophagy in MHC class I antigen presentation.Scand J Immunol. 2020 Nov;92(5):e12978. doi: 10.1111/sji.12978. Epub 2020 Oct 19. Scand J Immunol. 2020. PMID: 32969499 Free PMC article. Review.
-
Spotlight on TAP and its vital role in antigen presentation and cross-presentation.Mol Immunol. 2022 Feb;142:105-119. doi: 10.1016/j.molimm.2021.12.013. Epub 2021 Dec 29. Mol Immunol. 2022. PMID: 34973498 Free PMC article. Review.
Cited by
-
Bacterial Membrane Vesicles in Pneumonia: From Mediators of Virulence to Innovative Vaccine Candidates.Int J Mol Sci. 2021 Apr 8;22(8):3858. doi: 10.3390/ijms22083858. Int J Mol Sci. 2021. PMID: 33917862 Free PMC article. Review.
-
A cooperativity between virus and bacteria during respiratory infections.Front Microbiol. 2023 Nov 30;14:1279159. doi: 10.3389/fmicb.2023.1279159. eCollection 2023. Front Microbiol. 2023. PMID: 38098657 Free PMC article. Review.
-
Bacterial Membrane Vesicles: Physiological Roles, Infection Immunology, and Applications.Adv Sci (Weinh). 2023 Sep;10(25):e2301357. doi: 10.1002/advs.202301357. Epub 2023 Jun 25. Adv Sci (Weinh). 2023. PMID: 37357142 Free PMC article. Review.
-
Secretion of Salmonella Pathogenicity Island 1-Encoded Type III Secretion System Effectors by Outer Membrane Vesicles in Salmonella enterica Serovar Typhimurium.Front Microbiol. 2018 Nov 23;9:2810. doi: 10.3389/fmicb.2018.02810. eCollection 2018. Front Microbiol. 2018. PMID: 30532744 Free PMC article.
-
Revisiting Host-Pathogen Interactions in Cystic Fibrosis Lungs in the Era of CFTR Modulators.Int J Mol Sci. 2023 Mar 5;24(5):5010. doi: 10.3390/ijms24055010. Int J Mol Sci. 2023. PMID: 36902441 Free PMC article. Review.
References
-
- Swiatecka-Urban A., Moreau-Marquis S., Maceachran D. P., Connolly J. P., Stanton C. R., Su J. R., Barnaby R., O'Toole G. A., Stanton B. A. (2006) Pseudomonas aeruginosa inhibits endocytic recycling of CFTR in polarized human airway epithelial cells. Am. J. Physiol. Cell Physiol. 290, C862–C872 - PubMed
-
- MacEachran D. P., Ye S., Bomberger J. M., Hogan D. A., Swiatecka-Urban A., Stanton B. A., O'Toole G. A. (2007) The Pseudomonas aeruginosa secreted protein PA2934 decreases apical membrane expression of the cystic fibrosis transmembrane conductance regulator. Infect. Immun. 75, 3902–3912 - PMC - PubMed
-
- Wilkinson T. M., Donaldson G. C., Johnston S. L., Openshaw P. J., Wedzicha J. A. (2006) Respiratory syncytial virus, airway inflammation, and FEV1 decline in patients with chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 173, 871–876 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

