Polychlorinated biphenyls impair dibutyryl cAMP-induced astrocytic differentiation in rat C6 glial cell line
- PMID: 24251112
- PMCID: PMC3829991
- DOI: 10.1016/j.fob.2013.10.008
Polychlorinated biphenyls impair dibutyryl cAMP-induced astrocytic differentiation in rat C6 glial cell line
Abstract
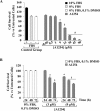
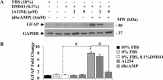
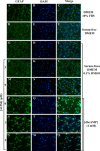
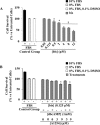
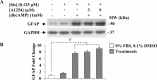
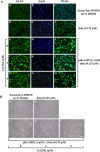
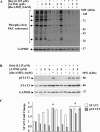
In the central nervous system, alteration of glial cell differentiation can affect brain functions. Polychlorinated biphenyls (PCBs) are persistent environmental chemical contaminants that exert neurotoxic effects in glial and neuronal cells. We examined the effects of a commercial mixture of PCBs, Aroclor1254 (A1254) on astrocytic differentiation of glial cells, using the rat C6 cell line as in vitro model. The exposure for 24 h to sub-toxic concentrations of A1254 (3 or 9 μM) impaired dibutyryl cAMP-induced astrocytic differentiation as showed by the decrease of glial fibrillary acidic protein (GFAP) protein levels and inhibition in change of cell morphology toward an astrocytic phenotype. The A1254 inhibition was restored by the addition of a protein kinase C (PKC) inhibitor, bisindolylmaleimide (bis), therefore indicating that PCBs disturbed the cAMP-induced astrocytic differentiation of C6 cells via the PKC pathway. The phosphorylation of signal transducer and activator of transcription 3 (STAT3) is essential for cAMP-induced transcription of GFAP promoter in C6 cells. Our results indicated that the exposure to A1254 (3 or 9 μM) for 24 h suppressed cAMP-induced STAT3 phosphorylation. Moreover, A1254 reduced cAMP-dependent phosphorylation of STAT3 requires inhibition of PKC activity. Together, our results suggest that PCBs induce perturbation in cAMP/PKA and PKC signaling pathway during astrocytic differentiation of glial cells.
Keywords: A1254, Aroclor 1254; Aroclor1254; Astrocytic differentiation; C6 glial cell line; CNS, central nervous system; CRE, cAMP responsive element; CREB, cAMP-response element binding protein; DAPI, 4′,6-diamidino-2-phenylindole; DMEM, Dulbecco’s Modified Eagle’s Medium; DMSO, dimethyl sulfoxide; GAPDH, glyceraldehyde-3-phosphate dehydrogenase; GFAP, glial fibrillary acidic protein; Glial fibrillary acidic protein (GFAP); MTT, 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide; NMDA, N-methyl-d-aspartate; PCBs, polychlorinated biphenyls; PKA, protein kinase A; PKC, protein kinase C; Protein kinase C (PKC); ROS, reactive oxygen species; STAT3, signal transducer and activator of transcription 3; Signal transducer and activator of transcription 3 (STAT3); TRE, CRE transcriptional response element; bis, 2-[1-(3-dimethylamino-propyl)indol-3-yl]-3-(indol-3-yl) maleimide; dbcAMP, N6,2′-O-dibutyryl cAMP; nNOS, neuronal nitric oxide.
Figures

Similar articles
-
Effects of polychlorinated biphenyls (PCBs) on expression of neurotrophic factors in C6 glial cells in culture.Neurotoxicology. 2007 Nov;28(6):1264-71. doi: 10.1016/j.neuro.2007.08.005. Epub 2007 Aug 19. Neurotoxicology. 2007. PMID: 17888513
-
cAMP-induced astrocytic differentiation of C6 glioma cells is mediated by autocrine interleukin-6.J Biol Chem. 2004 Apr 9;279(15):15441-7. doi: 10.1074/jbc.M311844200. Epub 2004 Jan 29. J Biol Chem. 2004. PMID: 14754894
-
Identification of calcium-dependent and -independent signaling pathways involved in polychlorinated biphenyl-induced cyclic AMP-responsive element-binding protein phosphorylation in developing cortical neurons.Neuroscience. 2002;115(2):559-73. doi: 10.1016/s0306-4522(02)00343-3. Neuroscience. 2002. PMID: 12421622
-
Dibutyryl cAMP- or Interleukin-6-induced astrocytic differentiation enhances mannose binding lectin (MBL)-associated serine protease (MASP)-1/3 expression in C6 glioma cells.Arch Biochem Biophys. 2018 Sep 1;653:39-49. doi: 10.1016/j.abb.2018.06.016. Epub 2018 Jun 28. Arch Biochem Biophys. 2018. PMID: 29963999
-
Cyclic AMP-mediated induction of the glial fibrillary acidic protein is independent of protein kinase A activation in rat C6 glioma.J Neurosci Res. 1997 May 15;48(4):324-33. J Neurosci Res. 1997. PMID: 9169858
Cited by
-
Mel1c Mediated Monochromatic Light-Stimulated IGF-I Synthesis through the Intracellular Gαq/PKC/ERK Signaling Pathway.Int J Mol Sci. 2019 Apr 4;20(7):1682. doi: 10.3390/ijms20071682. Int J Mol Sci. 2019. PMID: 30987295 Free PMC article.
-
Annurca Apple Polyphenol Extract Affects Acetyl- Cholinesterase and Mono-Amine Oxidase In Vitro Enzyme Activity.Pharmaceuticals (Basel). 2021 Jan 14;14(1):62. doi: 10.3390/ph14010062. Pharmaceuticals (Basel). 2021. PMID: 33466604 Free PMC article.
-
Neurotoxic Potential of Deoxynivalenol in Murine Brain Cell Lines and Primary Hippocampal Cultures.Toxins (Basel). 2022 Jan 10;14(1):48. doi: 10.3390/toxins14010048. Toxins (Basel). 2022. PMID: 35051025 Free PMC article.
-
Negative regulation of miR-1275 by H3K27me3 is critical for glial induction of glioblastoma cells.Mol Oncol. 2019 Jul;13(7):1589-1604. doi: 10.1002/1878-0261.12525. Epub 2019 Jun 18. Mol Oncol. 2019. PMID: 31162799 Free PMC article.
-
Vegetable Soups and Creams: Raw Materials, Processing, Health Benefits, and Innovation Trends.Plants (Basel). 2020 Dec 14;9(12):1769. doi: 10.3390/plants9121769. Plants (Basel). 2020. PMID: 33327480 Free PMC article. Review.
References
-
- Aschner M., Syversen T., Souza D.O., Rocha J.B., Farina M. Involvement of glutamate and reactive oxygen species in methylmercury neurotoxicity. Braz. J. Med. Biol. Res. 2007;40:285–291. - PubMed
-
- Tilson H.A., Jacobson J.L., Rogan W.J. Polychlorinated biphenyls and the developing nervous system: cross-species comparisons. Neurotoxicol. Teratol. 1990;12:239–248. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

