The role of nitric oxide signaling in food intake; insights from the inner mitochondrial membrane peptidase 2 mutant mice
- PMID: 24251118
- PMCID: PMC3830068
- DOI: 10.1016/j.redox.2013.10.003
The role of nitric oxide signaling in food intake; insights from the inner mitochondrial membrane peptidase 2 mutant mice
Abstract
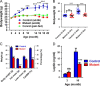
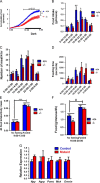
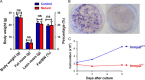
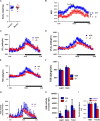
Reactive oxygen species have been implicated in feeding control through involvement in brain lipid sensing, and regulating NPY/AgRP and pro-opiomelanocortin (POMC) neurons, although the underlying mechanisms are unclear. Nitric oxide is a signaling molecule in neurons and it stimulates feeding in many species. Whether reactive oxygen species affect feeding through interaction with nitric oxide is unclear. We previously reported that Immp2l mutation in mice causes excessive mitochondrial superoxide generation, which causes infertility and early signs of aging. In our present study, reduced food intake in mutant mice resulted in significantly reduced body weight and fat composition while energy expenditure remained unchanged. Lysate from mutant brain showed a significant decrease in cGMP levels, suggesting insufficient nitric oxide signaling. Thus, our data suggests that reactive oxygen species may regulate food intake through modulating the bioavailability of nitric oxide.
Keywords: ADSC, adipose-derived stromal cells; AMPK, AMP-activated protein kinase.; AgRP, agouti related protein; CART, cocaine- and amphetamine-regulated transcript; CYC1, cytochrome c1; Energy expenditure; Food intake; GPD2, mitochondrial glycerol phosphate dehydrogenase; Immp2l; Immp2l, IMP2 inner mitochondrial membrane peptidase-like; Mutant mice; NO, nitric oxide; NOS, nitric oxide synthase; NPY, neuropeptide Y; Nitric oxide; POMC, pro-opiomelanocortin; ROS, reactive oxygen species; Superoxide; UCP2, uncoupling protein 2; cGMP, cyclic guanosine monophosphate.
Figures


Similar articles
-
Examination of bioenergetic function in the inner mitochondrial membrane peptidase 2-like (Immp2l) mutant mice.Redox Biol. 2014;2:1008-15. doi: 10.1016/j.redox.2014.08.006. Epub 2014 Aug 28. Redox Biol. 2014. PMID: 25460737 Free PMC article.
-
Mitochondrial peptidase IMMP2L mutation causes early onset of age-associated disorders and impairs adult stem cell self-renewal.Aging Cell. 2011 Aug;10(4):584-94. doi: 10.1111/j.1474-9726.2011.00686.x. Epub 2011 Apr 7. Aging Cell. 2011. PMID: 21332923 Free PMC article.
-
A mutation in the inner mitochondrial membrane peptidase 2-like gene (Immp2l) affects mitochondrial function and impairs fertility in mice.Biol Reprod. 2008 Apr;78(4):601-10. doi: 10.1095/biolreprod.107.065987. Epub 2007 Dec 19. Biol Reprod. 2008. PMID: 18094351
-
Leptin signaling, adiposity, and energy balance.Ann N Y Acad Sci. 2002 Jun;967:379-88. doi: 10.1111/j.1749-6632.2002.tb04293.x. Ann N Y Acad Sci. 2002. PMID: 12079865 Review.
-
Formation, signaling functions, and metabolisms of nitrated cyclic nucleotide.Nitric Oxide. 2013 Nov 1;34:10-8. doi: 10.1016/j.niox.2013.04.004. Epub 2013 Apr 28. Nitric Oxide. 2013. PMID: 23632125 Review.
Cited by
-
Suppression of Inner Mitochondrial Membrane Peptidase 2-Like (IMMP2L) Gene Exacerbates Hypoxia-Induced Neural Death Under High Glucose Condition.Neurochem Res. 2017 May;42(5):1504-1514. doi: 10.1007/s11064-017-2207-y. Epub 2017 Mar 18. Neurochem Res. 2017. PMID: 28316022
-
A single injection of neuropeptide QRFP in the lateral hypothalamus decreased food intake.J Psychopharmacol. 2025 Mar;39(3):254-264. doi: 10.1177/02698811241311454. Epub 2025 Feb 8. J Psychopharmacol. 2025. PMID: 39921588 Free PMC article.
-
Antioxidant Behavioural Phenotype in the Immp2l Gene Knock-Out Mouse.Genes (Basel). 2023 Aug 28;14(9):1717. doi: 10.3390/genes14091717. Genes (Basel). 2023. PMID: 37761857 Free PMC article.
-
Mitochondrial Processing Peptidases-Structure, Function and the Role in Human Diseases.Int J Mol Sci. 2022 Jan 24;23(3):1297. doi: 10.3390/ijms23031297. Int J Mol Sci. 2022. PMID: 35163221 Free PMC article. Review.
-
Nitric Oxide Exerts Basal and Insulin-Dependent Anorexigenic Actions in POMC Hypothalamic Neurons.Mol Endocrinol. 2016 Apr;30(4):402-16. doi: 10.1210/me.2015-1275. Epub 2016 Mar 1. Mol Endocrinol. 2016. PMID: 26930171 Free PMC article.
References
-
- Benani A., Troy S., Carmona M.C., Fioramonti X., Lorsignol A., Leloup C., Casteilla L., Penicaud L. Role for mitochondrial reactive oxygen species in brain lipid sensing: redox regulation of food intake. Diabetes. 2007;56:152–160. - PubMed
-
- Diano S., Liu Z.W., Jeong J.K., Dietrich M.O., Ruan H.B., Kim E., Suyama S., Kelly K., Gyengesi E., Arbiser J.L., Belsham D.D., Sarruf D.A., Schwartz M.W., Bennett A.M., Shanabrough M., Mobbs C.V., Yang X., Gao X.B., Horvath T.L. Peroxisome proliferation-associated control of reactive oxygen species sets melanocortin tone and feeding in diet-induced obesity. Nat. Med. 2011;17:1121–1127. - PMC - PubMed
-
- Choi Y.H., Furuse M., Okumura J., Denbow D.M. Nitric oxide controls feeding behavior in the chicken. Brain Res. 1994;654:163–166. - PubMed
-
- Khan M.S., Tachibana T., Hasebe Y., Masuda N., Ueda H. Peripheral or central administration of nitric oxide synthase inhibitor affects feeding behavior in chicks. Comp. Biochem. Physiol.. Part A Mol. Integr. Physiol. 2007;148:458–462. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

