The type of Aβ-related neuronal degeneration differs between amyloid precursor protein (APP23) and amyloid β-peptide (APP48) transgenic mice
- PMID: 24252227
- PMCID: PMC4046770
- DOI: 10.1186/2051-5960-1-77
The type of Aβ-related neuronal degeneration differs between amyloid precursor protein (APP23) and amyloid β-peptide (APP48) transgenic mice
Abstract
Background: The deposition of the amyloid β-peptide (Aβ) in the brain is one of the hallmarks of Alzheimer's disease (AD). It is not yet clear whether Aβ always leads to similar changes or whether it induces different features of neurodegeneration in relation to its intra- and/or extracellular localization or to its intracellular trafficking routes. To address this question, we have analyzed two transgenic mouse models: APP48 and APP23 mice. The APP48 mouse expresses Aβ1-42 with a signal sequence in neurons. These animals produce intracellular Aβ independent of amyloid precursor protein (APP) but do not develop extracellular Aβ plaques. The APP23 mouse overexpresses human APP with the Swedish mutation (KM670/671NL) in neurons and produces APP-derived extracellular Aβ plaques and intracellular Aβ aggregates.
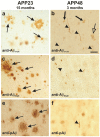
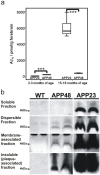
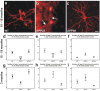
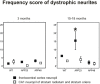
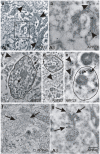
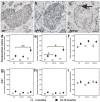
Results: Tracing of commissural neurons in layer III of the frontocentral cortex with the DiI tracer revealed no morphological signs of dendritic degeneration in APP48 mice compared to littermate controls. In contrast, the dendritic tree of highly ramified commissural frontocentral neurons was altered in 15-month-old APP23 mice. The density of asymmetric synapses in the frontocentral cortex was reduced in 3- and 15-month-old APP23 but not in 3- and 18-month-old APP48 mice. Frontocentral neurons of 18-month-old APP48 mice showed an increased proportion of altered mitochondria in the soma compared to wild type and APP23 mice. Aβ was often seen in the membrane of neuronal mitochondria in APP48 mice at the ultrastructural level.
Conclusions: These results indicate that APP-independent intracellular Aβ accumulation in APP48 mice is not associated with dendritic and neuritic degeneration but with mitochondrial alterations whereas APP-derived extra- and intracellular Aβ pathology in APP23 mice is linked to dendrite degeneration and synapse loss independent of obvious mitochondrial alterations. Thus, Aβ aggregates in APP23 and APP48 mice induce neurodegeneration presumably by different mechanisms and APP-related production of Aβ may, thereby, play a role for the degeneration of neurites and synapses.
Figures




Similar articles
-
Dispersible amyloid β-protein oligomers, protofibrils, and fibrils represent diffusible but not soluble aggregates: their role in neurodegeneration in amyloid precursor protein (APP) transgenic mice.Neurobiol Aging. 2012 Nov;33(11):2641-60. doi: 10.1016/j.neurobiolaging.2011.12.032. Epub 2012 Feb 2. Neurobiol Aging. 2012. PMID: 22305478
-
Selective vulnerability of different types of commissural neurons for amyloid beta-protein-induced neurodegeneration in APP23 mice correlates with dendritic tree morphology.Brain. 2006 Nov;129(Pt 11):2992-3005. doi: 10.1093/brain/awl176. Epub 2006 Jul 14. Brain. 2006. PMID: 16844716
-
Impact of amyloid β aggregate maturation on antibody treatment in APP23 mice.Acta Neuropathol Commun. 2015 Jul 4;3:41. doi: 10.1186/s40478-015-0217-z. Acta Neuropathol Commun. 2015. PMID: 26141728 Free PMC article.
-
Alzheimer's disease.Subcell Biochem. 2012;65:329-52. doi: 10.1007/978-94-007-5416-4_14. Subcell Biochem. 2012. PMID: 23225010 Review.
-
APP transgenic modeling of Alzheimer's disease: mechanisms of neurodegeneration and aberrant neurogenesis.Brain Struct Funct. 2010 Mar;214(2-3):111-26. doi: 10.1007/s00429-009-0232-6. Epub 2009 Nov 29. Brain Struct Funct. 2010. PMID: 20091183 Free PMC article. Review.
Cited by
-
Interactions of pathological proteins in neurodegenerative diseases.Acta Neuropathol. 2017 Aug;134(2):187-205. doi: 10.1007/s00401-017-1709-7. Epub 2017 Apr 11. Acta Neuropathol. 2017. PMID: 28401333 Free PMC article. Review.
-
Effect of AmyTrap, an amyloid-β binding drug, on Aβ induced mitochondrial dysfunction and tau phosphorylation in cultured neuroblastoma cells.Metab Brain Dis. 2020 Aug;35(6):923-931. doi: 10.1007/s11011-019-00520-2. Epub 2020 May 4. Metab Brain Dis. 2020. PMID: 32367269 Free PMC article.
-
Neuron Loss in Alzheimer's Disease: Translation in Transgenic Mouse Models.Int J Mol Sci. 2020 Oct 30;21(21):8144. doi: 10.3390/ijms21218144. Int J Mol Sci. 2020. PMID: 33143374 Free PMC article. Review.
-
Metabolic Characterization of Intact Cells Reveals Intracellular Amyloid Beta but Not Its Precursor Protein to Reduce Mitochondrial Respiration.PLoS One. 2016 Dec 22;11(12):e0168157. doi: 10.1371/journal.pone.0168157. eCollection 2016. PLoS One. 2016. PMID: 28005987 Free PMC article.
-
Deciphering the molecular profile of plaques, memory decline and neuron loss in two mouse models for Alzheimer's disease by deep sequencing.Front Aging Neurosci. 2014 Apr 16;6:75. doi: 10.3389/fnagi.2014.00075. eCollection 2014. Front Aging Neurosci. 2014. PMID: 24795628 Free PMC article.
References
-
- Alzheimer A. Über eine eigenartige Erkrankung der Hirnrinde. Allg Zschr Psych. 1907;64:146–148.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

