Establishing quantitative real-time quaking-induced conversion (qRT-QuIC) for highly sensitive detection and quantification of PrPSc in prion-infected tissues
- PMID: 24252329
- PMCID: PMC3893511
- DOI: 10.1186/2051-5960-1-44
Establishing quantitative real-time quaking-induced conversion (qRT-QuIC) for highly sensitive detection and quantification of PrPSc in prion-infected tissues
Abstract
Background: PrPSc, the only known constituent of prions, the infectious agents causing prion diseases, can be detected by real-time quaking-induced conversion (RT-QuIC). However, there is no efficient method to quantify the amount of PrPSc by RT-QuIC.
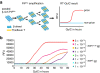
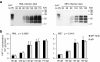
Results: Here we introduce quantitative RT-QuIC (qRT-QuIC) to quantify with high accuracy minute amounts of PrPSc in the brain and various peripheral tissues at levels far below detection by in vivo transmission. PrPSc is relatively resistant to treatment with proteinase K (PK). However, as there can also be a fraction of pathological PrP that is digested by PK, we use the term PrP27-30 to denote to the amount of PrPSc that can be detected by immunoblot after PK treatment. qRT-QuIC is based upon the quantitative correlation between the seeded amount of PrP27-30 and the lag time to the start of the conversion reaction detected by RT-QuIC. By seeding known amounts of PrP27-30 quantified by immunoblot into qRT-QuIC a standard calibration curve can be obtained. Based on this calibration curve, seeded undetermined amounts of PrP27-30 can be directly calculated. qRT-QuIC allowed to quantify PrP27-30 concentrations at extremely low levels as low as 10-15.5 g PrP27-30, which corresponds to 0.001 LD50 units obtained by in vivo i.c. transmission studies. We find that PrP27-30 concentration increases steadily in the brain after inoculation and can be detected at various time points during the incubation period in peripheral organs (spleen, heart, muscle, liver, kidney) in two experimental scrapie strains (RML, ME7) in the mouse.
Conclusions: We suggest that an automatic quantitative system to measure disease progression as well as prion contamination of organs, blood and food product is feasible. Moreover, the concept of qRT-QuIC should be applicable to measure other disease-associated proteins rich in β-pleated structures (amyloid) that bind ThT and that show seeded aggregation.
Figures



References
-
- Pan KM, Baldwin M, Nguyen J, Gasset M, Serban A, Groth D, Mehlhorn I, Huang Z, Fletterick RJ, Cohen FE. Conversion of alpha-helices into beta-sheets features in the formation of the scrapie prion proteins. Proc Natl Acad Sci USA. 1993;1:10962–10966. doi: 10.1073/pnas.90.23.10962. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

